
(a)
Interpretation:
The constitutional isomer, which is formed greatest yield in the given reaction should be given.
Concept introduction:
Dehydration reaction:
Removal of water molecule from the reaction when the alcohol is treated with strong acid like sulfuric acid is known as dehydration reaction, for example
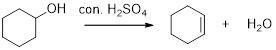
Alcohols are react with acids like hydrochloric acid or hydrobromic to yields the corresponding carbocation intermediates, this carbocation intermediate undergoes elimination reaction to give a corresponding
The stability of carbocation is given below,
Tertiary carbocation is more stable than the secondary and primary.
Cis–trans isomerism (or) geometric isomerism or configurational isomerism:
The
The functional groups are in opposite to each other in the carbon chain is called trans isomer.
Two similar functional groups are in same side which is called as Z-isomer.
Two similar functional groups are opposite side which is called as E-isomer.
Stereoisomers: same molecular formula and sequence of bonded atoms (constitution), but differ in the three-dimensional orientations of their atoms in space.
(b)
Interpretation:
The stereoisomer, which is formed greatest yield in the given reaction should be given.
Concept introduction:
Dehydration reaction:
Dehydration reaction:
Removal of water molecule from the reaction when the alcohol is treated with strong acid like sulfuric acid is known as dehydration reaction, for example
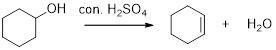
Alcohols are react with acids like hydrochloric acid or hydrobromic to yields the corresponding carbocation intermediates, this carbocation intermediate undergoes elimination reaction to give a corresponding alkene as a product.
The stability of carbocation is given below,
Tertiary carbocation is more stable than the secondary and primary.
Cis–trans isomerism (or) geometric isomerism or configurational isomerism:
The functional groups are in the same side of the carbon chain is called cis isomer.
The functional groups are in opposite to each other in the carbon chain is called trans- isomer.
Two similar functional groups are in same side which is called as Z-isomer.
Two similar functional groups are opposite side which is called as E-isomer.
Stereoisomers: same molecular formula and sequence of bonded atoms (constitution), but differ in the three-dimensional orientations of their atoms in space.
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Organic Chemistry (8th Edition)
- Optical active compound 1-chloro-3-methylcyclopentane was reacting withpotassium t-butoxide in t-butanol. Two alkene products were obtained. Themain product was optically active, and the secondary product was notoptically active. What are the two products?arrow_forwardConsider the reaction below: D D Br2 The final product of this reaction has [Select] form a total of [Select] stereocenter(s), and stereoisomer(s).arrow_forward1. Consider the reaction scheme below NBS hv a. Draw the major regioisomer resulting from monobromination of cyclohexene with N-bromosuccinimide. If this major regioisomer is produced as a mixture of stereoisomers, draw each stereoisomer and indicate the relative amount of each stereoisomer (That is, if one stereoisomer is formed preferentially label it is as "major." If both are formed in equal amounts label each as "50%"). b. Draw an arrow pushing reaction mechanism for this reaction. You should draw an initiation step, two propagation steps, and two possible termination steps for the radical chain reaction and clearly label which step is which. Your mechanism can use Br2 as the source of bromine radicals in the initiation step that will be present as a minor, low concentration impurity in the NBS. 36°Farrow_forward
- Draw all stereoisomers formed in each reaction. to [1] CH3LI а. [2] H2O 1m( Culi b. [1] [2] H2O [1] LIAIH4 С. с. [2] H2O [1] mCРBA d. [2] MgBr [3] H2O ..||arrow_forwardDraw all stereoisomers formed in the reaction shown. Use wedges and dashes for tetrahedral stereogenic centers, if applicable. NH₂ HOACarrow_forwardCompare the physical properties of the three stereoisomers of 1,3-dimethylcyclopentane. a.How do the boiling points of A and B compare? What about those of A and C? b. Characterize a solution of each of the following as optically active or optically inactive: pure A; pure B; pure C; an equal mixture of A and B; an equal mixture of A and C. c.A reaction forms a 1:1:1 mixture of A, B, and C. If this mixture is distilled, how many fractions would be obtained? Which fractions would be optically active and which would be optically inactive?arrow_forward
- What are the products of the following reaction? a. How many stereoisomers of each product could be obtained?arrow_forward2. a. b. C. Predict the products of the reactions below. Show stereochemistry, using wedges and dashes, in the products and draw both enantiomers. 1. Hg(OAc)2, H₂O 2. NaBH4 1. BH3 2. H₂O2, NaOH H₂ Pd/Carrow_forwardWhich of the molecules A through D shown below will give mixtures of enantiomers upon reaction with HCI? C&H5 CeHs A B C D O 1, Molecules B and C. O 2, Molecules A and B. O 3, Molecules C and D. 0 4. Molecules A and C.arrow_forward
- What stereoisomers are obtained from the following reactions? a. 1-butene + H2O + H2SO4 b. cyclohexene + HBr c. cis-3-heptene + Br2 d. trans-3-hexene + Br2arrow_forward2.) Which is a pair of enantiomers? CH3 H3C CI CH;CH2CH3 CH3 CH3 CH;CH;CH3 H3C CI CH;CH2CH; II II CH,CI CH;CH2CH3 H3C CI CH;CH2CH3 IV V A. I& II and III & IV В. I & II С. II & IV D. IV & Varrow_forwardDraw the cycloalkene that would react with the reagent given to account for the product formed. ? + HCI CH3 • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





