
Prescott's Microbiology
11th Edition
ISBN: 9781260211887
Author: WILLEY, Sandman, Wood
Publisher: McGraw Hill
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 10.7, Problem 1MI
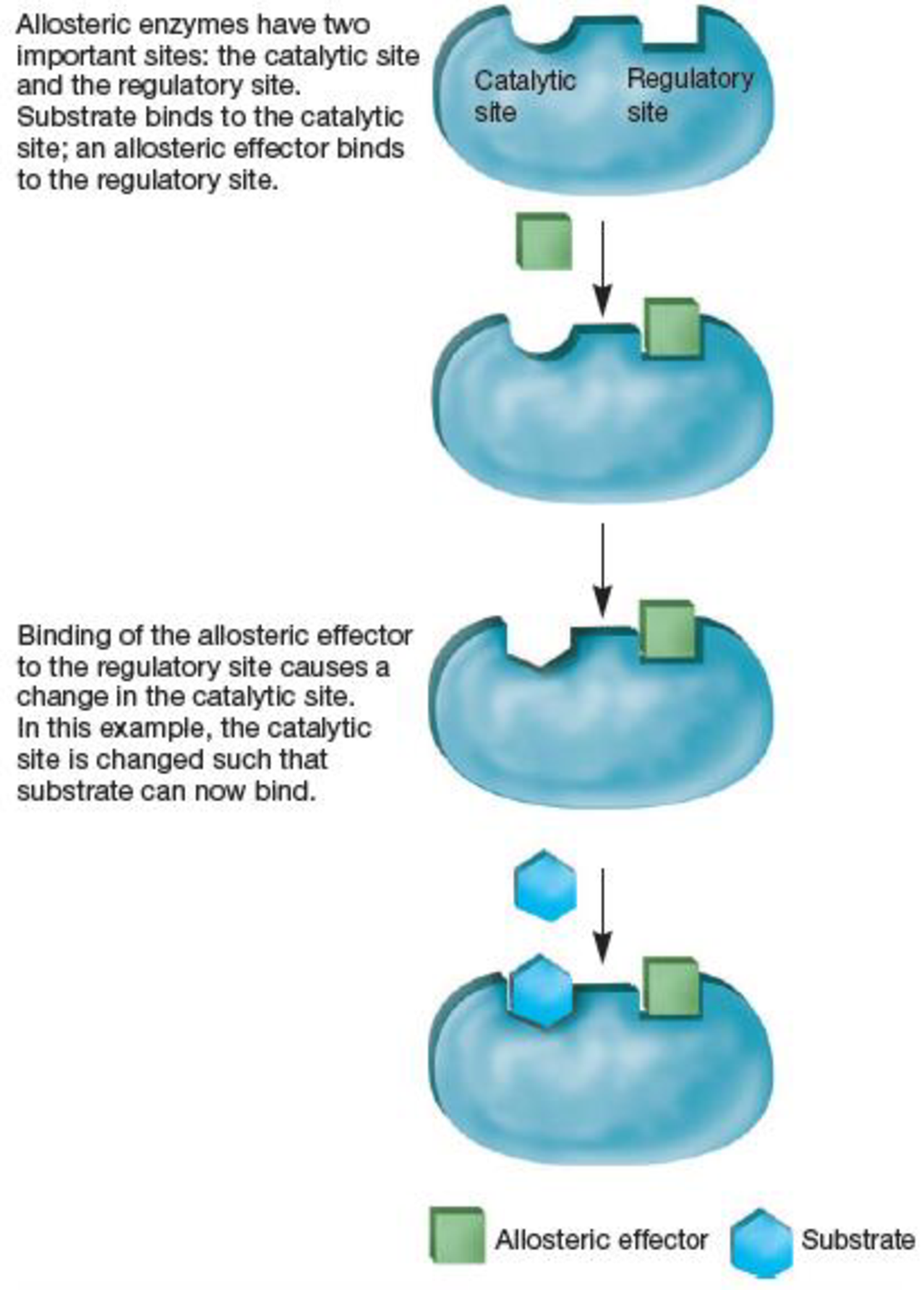
Figure 10.19 Allosteric Regulation. The structure and function of an allosteric enzyme.
Is this allosteric effector a positive or negative effector? Is this true of all allosteric effectors?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
There are several types of enzyme regulators which can either be positive ("turn on") or negative ("turn off") regulators. Why would you ever want to "turn off" an enzyme?
Why does CTP favor the T state while ATP favors the R state? (in reference to CTPase).
Do you think the hydrolysis GTP and ATP are similar? Explain why or why not?
Chapter 10 Solutions
Prescott's Microbiology
Ch. 10.1 - Figure 10.2 The Relationship of G to the...Ch. 10.1 - Prob. 1CCCh. 10.1 - Prob. 2CCCh. 10.1 - Prob. 3CCCh. 10.1 - Prob. 4CCCh. 10.2 - Why is ATP called a high-energy molecule? How is...Ch. 10.2 - Describe the energy cycle and ATPs role in it....Ch. 10.3 - Prob. 1MICh. 10.3 - Prob. 2MICh. 10.4 - Figure 10.6 Electron Movement and Reduction...
Ch. 10.4 - How is the direction of electron flow between...Ch. 10.4 - When electrons flow from the NAD+/NADH conjugate...Ch. 10.4 - Which among the following would be the best...Ch. 10.4 - In general terms, how is G related to E0? What is...Ch. 10.4 - Name and briefly describe the major electron...Ch. 10.6 - Will an enzyme with a relatively high Km have a...Ch. 10.6 - Prob. 2MICh. 10.6 - Prob. 1CCCh. 10.6 - Prob. 2CCCh. 10.6 - How does enzyme activity change with substrate...Ch. 10.6 - What special properties might an enzyme isolated...Ch. 10.6 - What are competitive and noncompetitive...Ch. 10.6 - How are enzymes and ribozymes similar? How do they...Ch. 10.7 - Figure 10.19 Allosteric Regulation. The structure...Ch. 10.7 - Prob. 2MICh. 10.7 - Define the terms metabolic channeling and...Ch. 10.7 - Define allosteric enzyme and allosteric effector.Ch. 10.7 - Prob. 3CCCh. 10.7 - Prob. 4CCCh. 10.7 - Prob. 5CCCh. 10 - Prob. 1RCCh. 10 - Prob. 2RCCh. 10 - Prob. 3RCCh. 10 - Examine the structures of macromolecules in...Ch. 10 - Examine the branched pathway shown here for the...Ch. 10 - Prob. 3AL
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Kinesin movement is dependent on GTP hydrolysis. True Falsearrow_forwardhow to read this table? like for example, just by looking at the table, it was said that leptin has 16 kda with 15% gel. How did they able to determine that leptin needs 15% gel?arrow_forward6)During the epinephrine signaling pathway we learned about in class, the enzyme glycogen phosphorylase is regulated by: Zymogen activation Competitive inhibition Allosteric regulation Covalent modificationarrow_forward
- if the reaction pH drops significantly (pH=2 for example), how would reaction catalyzed be serine proteases be affected? why? (assumin that the rest of the protein structure remain intact)arrow_forwarddrop down choices: - functional - non functional - no transcriptionarrow_forwardBIOMOLECULES - MULTIPLE CHOICE - Please answer properly QUESTION : Major controls of de novo AMP synthesis include: 1. allosteric inhibition by GMP allosteric inhibition by AMP 3. availability of PRPP 4. stimulation by GTP A. 1, 2, 3, and 4 B. 1, 2, and 3 C. 2 and 4 D. 1 and 3arrow_forward
- True or False 1. Predominant nucleotides during protein synthesis are the GTPs. 2. Predominant nucleotides during protein synthesis are the GTPs. 3. The nucleoside monophosphates are seen in metabolic pathways because their phosphoric anhydride bonds can be hydrolyzed off for energy.arrow_forwardThe phosphorylation and dephosphorylation of proteins is a vital means of regulation. Protein kinases attach phosphoryl groups, whereas only a phosphatase will remove the phosphoryl group from the target protein. What is the energy cost of this means of covalent regulation?arrow_forwardThe allosteric regulation of ATCase by CTP is an example of: 1. Negative homotropic allostery 2. Positive homotropic allostery 3. Negative heterotropic allostery 4. Positive heterotropic allosteryarrow_forward
- Which factor has NOT been shown to play a role in determining the specificity of protein kinases? a. protein tertiary structure b. protein quaternary structure c. primary sequence at phosphorylation site d. disulfide bonds near the phosphorylation site e. residues near the phosphorylation sitearrow_forwardhow allosteric regulation is fundamentally different from competitive/uncompetitive/mixed inhibition and why Michaelis-Menten and Lineweaver-Burk analysis cannot be used for allosteric enzymes.arrow_forwardLigand binding and response. The following question involves the ligand binding to a receptor and the receptor's response to that ligand. What ligand concentration would be required for a full agonist with a KD of 8 nM to achieve a response of 0.75?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning
Nervous System - Get to know our nervous system a bit closer, how does it works? | Neurology; Author: FreeMedEducation;https://www.youtube.com/watch?v=6O-0CVAgaEM;License: Standard youtube license