
Pushing Electrons
4th Edition
ISBN: 9781133951889
Author: Weeks, Daniel P.
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 30EQ
Derive Lewis structures for the compounds below.
Azobenzene
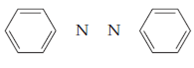
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Please correct answer and don't used hand raiting
this is an induced approximation. Can
u draw the mechanism ?
요
H
NaOH
1-1
OH
Please correct answer and don't used hand raiting
Chapter 1 Solutions
Pushing Electrons
Ch. 1 - 1. Hydrogen is a Group I element and each...Ch. 1 - Methanol has the molecular formula CH4O. Its...Ch. 1 - 3. The skeleton of chloromethane is...Ch. 1 - 4. Methanol’s skeleton is
Connecting all bonded...Ch. 1 - 5. The structure for chloromethane is
It...Ch. 1 - Prob. 6EQCh. 1 - 7. Dimethyl ether
No. of electrons in...Ch. 1 - Methylamine (CH5N) No. of electrons in structure...Ch. 1 - Methanethiol (CH4S) No. of electrons in structure...Ch. 1 - Methylal (C3H8O2) No. of electrons in structure...
Ch. 1 - Prob. 11EQCh. 1 - Adding electrons to the skeleton by making single...Ch. 1 - This is done by removing an unshared pair from...Ch. 1 - Prob. 14EQCh. 1 - Prob. 15EQCh. 1 - Prob. 16EQCh. 1 - The skeleton of acetyl chloride is . Write the...Ch. 1 - Three constitutional isomers exist for the formula...Ch. 1 - A number of constitutional isomers exist for the...Ch. 1 - Using the method outlined above, derive the...Ch. 1 - Prob. 21EQCh. 1 - Prob. 22EQCh. 1 - Prob. 23EQCh. 1 - Prob. 24EQCh. 1 - The skeleton of benzyldimethylamine is
The...Ch. 1 - The skeleton is benzaldoxime is The number of...Ch. 1 - Prob. 27EQCh. 1 - Derive Lewis structures for the compounds below....Ch. 1 - Prob. 29EQCh. 1 - Derive Lewis structures for the compounds below....Ch. 1 - Prob. 31EQCh. 1 - Derive Lewis structures for the compounds below....Ch. 1 - The Lewis structure of acetone is Circling the...Ch. 1 - Chloromethane has the Lewis...Ch. 1 - In the Lewis structure for chloromethane, the...Ch. 1 - Prob. 36EQCh. 1 - The oxygen atom in acetone possesses ____ unshared...Ch. 1 - Nitrobenzene has the skeleton
The number of...Ch. 1 - Prob. 39EQCh. 1 - Compute and add on the formal charges I these...Ch. 1 - Prob. 41EQCh. 1 - Prob. 42EQCh. 1 - Prob. 43EQCh. 1 - Prob. 44EQCh. 1 - Prob. 45EQCh. 1 - Prob. 46EQCh. 1 - Prob. 47EQCh. 1 - Compute and add on the formal charges in these...Ch. 1 - Prob. 49EQCh. 1 - Prob. 50EQCh. 1 - The n-propyl cation can be formed from a molecule...Ch. 1 - Prob. 52EQCh. 1 - Prob. 53EQCh. 1 - Methanol, CH3OH, is a compound in which the formal...Ch. 1 - When a proton becomes bonded to diethyl ether, by...Ch. 1 - Tetrahydrofuran has the structure
When a proton...Ch. 1 - Prob. 57EQCh. 1 - Prob. 58EQCh. 1 - The structure of pyridine is
When a proton...Ch. 1 - The carbon atom owns one electron from each of ...Ch. 1 - The n-butyl anion can be formed from When the CLi...Ch. 1 - The isobutyl anion can be formed from When the CNa...Ch. 1 - Prob. 63EQCh. 1 - Ethanol, , is a compound in which the formal...Ch. 1 - The loss of a proton attached to the oxygen atom...Ch. 1 - A very strong base can remove a proton from...Ch. 1 - Prob. 67EQCh. 1 - Prob. 68EQCh. 1 - Prob. 69EQCh. 1 - The homolysis of the OO bond in diacetyl peroxide...Ch. 1 - Prob. 71EQCh. 1 - Prob. 72EQCh. 1 - Prob. 73EQCh. 1 - Prob. 74EQCh. 1 - Prob. 75EQCh. 1 - Heterolytic cleavage of the CO bond to yield a...Ch. 1 - Prob. 77EQCh. 1 - Prob. 78EQCh. 1 - Prob. 79EQCh. 1 - Prob. 80EQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Please correct answer and don't used hand raitingarrow_forwardPlease correct answer and don't used hand raitingarrow_forward(a) The following synthesis of the molecule shown in the circle has a major problem. What is this problem? (2 pts) 1) HBr (no peroxides) 2) H- NaNH2 Br 3) NaNH, 4) CH3Br 5) H2, Pd (b) Starting with the molecule shown below and any other materials with two carbons or less, write out an alternate synthesis of the circled molecule. More than one step is needed. Indicate the reagent(s) and the major product in all the steps in your synthesis. (5 pts) 2024 Fall Term (1) Organic Chemistry 1 (Lec) CHEM 22204 02[6386] (Hunter College) (c) Using the same starting material as in part (b) and any other materials win two carpons or less, write out syntheses of the circled molecules shown below. More than one step is needed in each case. Indicate the reagent(s) and the major product in all the steps in your synthesis. You may use reactions and products from your synthesis in part (b). (5 pts)arrow_forward
- alt ons for Free Response Questions FRQ 1: 0/5 To spectrophotometrically determine the mass percent of cobalt in an ore containing cobalt and some inert materials, solutions with known [Co?) are prepared and absorbance of each of the solutions is measured at the wavelength of optimum absorbance. The data are used to create a calibration plot, shown below. 0.90- 0.80- 0.70 0.60 0.50 0.40- 0.30 0.20- 0.10- 0.00- 0.005 0.010 Concentration (M) 0.015 A 0.630 g sample of the ore is completely dissolved in concentrated HNO3(aq). The mixture is diluted with water to a final volume of 50.00 ml. Assume that all the cobalt in the ore sample is converted to Co2+(aq). a. What is the [Co2] in the solution if the absorbance of a sample of the solution is 0.74? 13 ✗ b. Calculate the number of moles of Co2+(aq) in the 50.00 mL solution. 0.008 mols Coarrow_forwardPlease correct answer and don't used hand raitingarrow_forwardCloso-boranes and arachno-boranes are structures that exhibit B-B, B-H-B, and B-H bonds. Correct?arrow_forward
- Indicate why boron hydrides cannot form large linear or planar structures.arrow_forwardNido-boranes are structures with the molecular formula BnHn+4 that exhibit B-B, B-H-B and B-H bonds. Correct?arrow_forward8:07 AM Wed Dec 18 Final Exam 2024 copy Home Insert Draw Page Layout Formulas Data Review AA 田 General A G fx Alexis Cozort ☑ ⚫ 61% A B D E F H K M N P R S T U 3+ 10 125 mM that yielded peak heights of Aa = 9 1-(a)A sample solution was examined under XRF to quantify the analyte Ce³+. Find the response factor F, when standardized concentration of analyte [Ce³+]A = concentration of internal standard S i.e. [In³*]s = 151 mM was spiked with standardized 1600 and As = 3015 respectively? 11 12 (i)Define F, F = Aa As [A] [S] + X 13 (*Define with variables) 4000 14 15 (ii)Calculate F, F = numeral (You will use the F value in part 1-(b) below) As 16 (*Calculate with numerals) 17 18 1-(b)To determine the unknown conc of analyte [Ce³+], a volume of 15 mL of internal standard S having a concentration [In³+]s = 0.264 M 19 20 was added to 45 mL of unknown, and the mixture was diluted to 100 mL in a volumetric flask. XRF analysis yielded a spectrum, Figure-1, where peak heights A and As are…arrow_forward
- All structural types of Boron hydrides exhibit B-B, B-H-B and B-H bonds. Correct?arrow_forwardN-nitrosodimethylamine (NDMA) is a suspected carcinogen that can form via reactions between dimethylamine (DMA) and monochloramine (NH2Cl). The relevant elementary reactions and the corresponding rate constants are as shown below. Reaction Rate constant (M¹s¹) DMA + NH2Cl = DMCA + NH3 k =1.4×10-1, kr = 5.83×10-3 1.28×10-3 DMA + NH2Cl → UDMH UDMH + NH2Cl → NDMA -> 1.11×10-1 If the initial concentrations of DMA and NH2Cl are given, you should be able to predict the concentrations of all species at any given reaction time. Please write down the rate equations for DMA, NH2C1, DMCA, UDMH and NDMA.arrow_forwardYou wish to add enough NaOCl (sodium hypochlorite) to a 150 m³ swimming pool to provide a dose of 5.0 mg/L TOTOCI as Cl2. (a) How much NaOCI (kg) should you add? (Note: the equivalent weight of NaOCl is based on the reaction: NaOCl + 2H + 2 e→CI + Na +H₂O.) (10 pts) (atomic weight: Na 23, O 16, C1 35.5) (b) The pH in the pool after the NaOCl addition is 8.67. To improve disinfection, you want at least 90% of the TOTOCI to be in the form of HOCI (pKa 7.53). Assuming that HOCI/OCI is the only weak acid/base group in solution, what volume (L) of 10 N HCl must be added to achieve the goal? (15 pts) Note that part a) is a bonus question for undergraduate students. If you decide not to work on this part of the question, you many assume TOTOCI = 7×10-5 M for part b).arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
INTRODUCTION TO MOLECULAR QUANTUM MECHANICS -Valence bond theory - 1; Author: AGK Chemistry;https://www.youtube.com/watch?v=U8kPBPqDIwM;License: Standard YouTube License, CC-BY