
Concept explainers
Methanol,
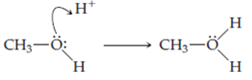
This time we use the curved arrow to signify bond making. Now a pair of unshared electrons on oxygen is pushed toward the region between the oxygen atom and the hydrogen ion. It becomes an
In the resulting structure the oxygen atom owns one electron from each of ____ shared pairs and two electrons from ____ unshared pair. The total number of electrons that belong to the oxygen atom is ____. Oxygen is a Group ____ element. Since the number of electrons that the oxygen atom owns in this structure is one fewer that it would have in the neutral, unbonded state, the charge on oxygen is ____. The correct Lewis structure for the conjugate acid of methanol is
_________________________
Once again charge is conserved. Joining a neutral molecule and a cation must yield a cation. Again the formal charge distribution on the resulting ion is predictable from the arrow. Electrons are pushed away from the oxygen atom, leaving it with a positive charge. Electrons are pushed toward the hydrogen ion, neutralizing its erstwhile positive charg.
Trending nowThis is a popular solution!

Chapter 1 Solutions
Pushing Electrons
- What is the formal charge on the indicated atom in each of the following species? (a) sulfur in SO2 (b) nitrogen in N2H4 (c) each oxygen atom in ozone, O3arrow_forwardChloromethane has the Lewis structure _______________________________ The carbon atom is sharing 4 electron pairs. In each shared pair the carbon atom “owns” 1 electron. The number of electrons that “belong” to carbon is ___. Carbon, being a Group ___ element would have 4 , outer shell electrons in the unbonded, neutral state. Therefore, the carbon atom in chloromethane has a formal charge of zero.arrow_forwardDetermine the formal charge on each atom in the following molecules or ions: (a) SCO (b) HCO2 (formate ion) (c) CO32 (d) HCO2H (formic acid)arrow_forward
- Draw two different Lewis structures for ClO3-, one which all atoms follow the octet rule and one where the chlorine has an expanded octet. Determine the formal charge on all atoms for each structure. Which do you think is the better Lewis structure? Explain your reasoning.arrow_forwardFor the NO3-1 anion, what are the formal charges on each atom?arrow_forward5.) Nitrous acid, HNO2, has 2 oxygens bonded to the central nitrogen atom. The order of the atoms is H O1 N O2 (note the numbers - they mean oxygen #1 and oxygen #2) What are the formal charges on each oxygen as well as the nitrogen atom? Group of answer choices oxygen #1 is 0 and oxygen #2 is 0 and nitrogen is 0 oxygen #1 is -1 and oxygen #2 is 0 and nitrogen is +1 oxygen #1 is +1 and oxygen #2 is -1 and nitrogen is 0 oxygen #1 is 0 and oxygen #2 is -1 and nitrogen is +1 oxygen #1 is +1 and oxygen #2 is -1 and nitrogen is +1 oxygen #1 is 0 and oxygen #2 is +1 and nitrogen is -1arrow_forward
- For the carbonate ion (CO32-), there are 3 resonance structures. For any given structure what would the formal charge be for an oxygen that has a single bond to the central carbon atom?arrow_forwardDetermine the formal charge on each atom in the ion H3O+ ?arrow_forwardCalculate the formal charge on the nitrogen atom in ammonia, NH3; in the ammonium ion, NH4+; and in the amide ion, NH2-.arrow_forward
- How many covalent bonds are there in ClO2? Assume Cl will expand its octet to minimize formal charges.arrow_forwardP3- ion is a linear ion composed of 3 phosphorus atoms. Draw 3 resonance structures of this ion. Show the individual formal charges of all atoms. Indicate which structure is the best one.arrow_forwardIn N2O, nitrogen is the central atom and the oxygen atom is terminal. In OF2, however, oxygen is the central atom. Use formal charges to explain why.arrow_forward

 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning





