
Concept explainers
Interpretation:
The pentose phosphate pathway for degrading sugars (see Problem 29-21) is the conversion of ribose 5-phosphate to glyceraldehyde 3-phosphate.
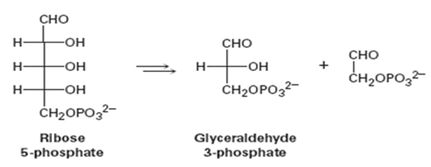
Concept introduction:
Reactions that make and break C-C-Bonds
Reactions that make and break carbon-carbon bonds form the basis of both degradative and biosynthetic
The breakdown of glucose to CO2 involves give such cleavages where as its synthesis involves the reverse process such reactions, considered from the synthetic direction involve addition of a nucleophilic carbanion to an electrophilic carbon atom. The most common electrophilic carbon atoms in such reactions are the sp2 hybridized carbonyl atoms of
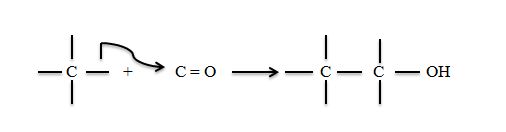
Stabilized carbanions must be generated to add to these electrophilic centres. Three examples are the aldol condensation (catalyzed eg by aldolase) claisen ester condensation (citrate synthase) and the decarboxylation of β-keto acids. (isocitrate dehydrogenase and fatly acid synthase).
Want to see the full answer?
Check out a sample textbook solution
Chapter 29 Solutions
Organic Chemistry
- 0 What are the structural differences between vitamin C and sugars? Do these structural differences play a role in the susceptibility of this vitamin to air oxidation?arrow_forwardIf an enzyme-catalyzed reaction has a high rate at low pH and low rate at higher pH, this implies that a group on either the enzyme or the substrate must be for an efficient reaction. leaving group oxidoreductase coenzymes O protonated deprotonated The compound that consists of deoxyribose linked by an N-glycosidic bond to N-9 of guanine is: adenylate deoxyguanosine guanosine nucleotide guanylatearrow_forwardStearic acid is the common name for the C18 straight-chain, saturated fatty acid. Draw the structure of the fatty acyl-CoA that this forms (you don't have to draw out all of the atoms in coenzyme A; you can represent it as S-, and then show all of the reactions). Draw & identify all of the compounds formed when the eighteen carbon fat is converted to two molecules of acetyl-CoA and the fourteen-carbon fat via two turns of the fatty acid spiral. List the enzymes and coenzymes that are part of the pathway.arrow_forward
- Which one of these molecules is a naturally occuring sugar?arrow_forwardTPP is a coenzyme for transketolase, the enzyme that catalyzes the conversion of a ketopentose (xylulose- 5-phosphate) and an aldopentose (ribose-5-phosphate) to an aldotriose (glyceraldehyde-3-phosphate) anda ketoheptose (sedoheptulose-7-phosphate). Notice that the total number of carbons in the reactants and products is the same (5 + 5 = 3 + 7). Propose a mechanism for this reaction.arrow_forwardClassify each compound as a reducing or nonreducing sugar.arrow_forward
- The enzyme 6-phosphogluconate dehydrogenase is part of the pentose pathway for glucose oxidation. What enzyme that is involved in glucose oxidation by the citric acid cycle has a very similar reaction mechanism to 6-phosphogluconate dehydrogenase? A) isocitrate dehydrogenase B) alpha-ketoglutarate dehydrogenase C) succinate dehydrogenase D) malate dehydrogenase E) pyruvate dehydrogenase (99+ RATI aarrow_forwardHow can glucose be utilized to produce ribose for RNA synthesis?arrow_forwardThe rate-limiting step is a metabolic pathway is the slowest step which determines the overall rate of the other reactions in the pathway. In glycolysis, the rate limiting step is a phosphorylation reaction where phosphofructokinase (PFK-1) catalyzes the reaction fructose-6-bisphosphate -> fructose-1,6-bisphosphate, the same step in gluconeogenesis. Select one: The statement is FALSE. The statement is TRUE.arrow_forward
- The enzyme that catalyzes this reaction is a(n) H 1000 CH3 Acetaldehyde Otransferase NADH + H+ ligase Olyase oxidoreductase hydrolase alcohol dehydrogenase NAD+ OH CH2 CH3 Ethanolarrow_forwardName: 1. Draw out the mechanism for the coupling of two glucose molecules to make maltose. CH₂OH CH₂OH НО OH ОН ОН НО OH ОН ОН 2. Sugar molecules are capable of greater structural diversity than proteins. What is the primary reason for this?arrow_forwardQuestion 5. The biosynthesis of fatty acids involves an enzyme-catalyzed hydrogenation of an a,ß-unsaturated thioester to the saturated fatty acid intermediate mediated by NADPH+. However, the reverse oxidation reaction during fatty acid metabolism requires a different cofactor FAD. Fatty Acid Biosynthesis a) Draw the structure of FAD. H flocon SCOA H SR + FAD - FADH₂ hydrogenation (reduction) NADPH+, H+ dehydrogenation (oxidation) FAD Fatty Acid Metabolism b) Draw a detailed mechanism of the dehydrogenation of fatty acyl-CoA by FAD (you can abbreviate AD as R). H SRarrow_forward

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

