
Concept explainers
Compound Y
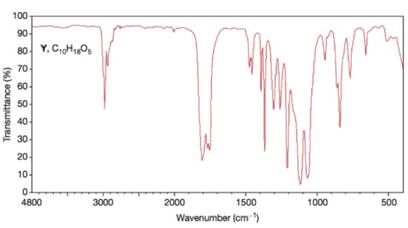
Figure 1 The IR spectrum of compound Y (Problem 34)
Want to see the full answer?
Check out a sample textbook solution
Chapter FRP Solutions
Organic Chemistry
Additional Science Textbook Solutions
General, Organic, and Biological Chemistry (3rd Edition)
Fundamentals of Heat and Mass Transfer
Chemistry For Changing Times (14th Edition)
Organic Chemistry (8th Edition)
- Compound I (C11H14O2) is insoluble in water, aqueous acid, and aqueous NaHCO3, but dissolves readily in 10% Na2CO3 and 10% NaOH. When these alkaline solutions are acidified with 10% HCl, compound I is recovered unchanged. Given this information and its 1H-NMR spectrum, deduce the structure of compound I.arrow_forward07) The proton NMR spectrum of a compound with formula C5H12O2 is shown below. THE The carbon-13 NMR spectrum has three peaks, respectively at 15 ppm, 63 ppm and 95 ppm. Based on this information draw the structure of this compound.arrow_forwardA compound with molecular formula C3H8O produces a broad signal between 3200 and 3600 cm³¹ in its IR spectrum and produces two signals in its 13C NMR spectrum. Draw the structure of the compound. Draw Your Solutionarrow_forward
- Compound 2 has molecular formula C6H12. It shows three signals in the 1H-NMR spectrum, one at 0.96 ppm, one at 2.03 ppm, and one at 5.33 ppm. The relative integrals of these three signals are 3, 2, and 1, respectively. Provide structure for compound 2, explain how you reached your conclusion.arrow_forwardDeduce the structure of a compound with molecular formula C5H100 that exhibits the following ¹H and ¹³C NMR spectra. IH NMR CNMR 150 10 Structure A Structure B Structure C Structure D 24 1C 100 B 2H H 20 50 10 D 311arrow_forwardGiven the following 13C NMR signals, construct a structure for the unknown compounds. A. Molecular formula: C6H14O DEPT-135 (positive): 14.1 δ DEPT-135 (negative): 22.7 δ, 25.3 δ, 31.8 δ, 32.2 δ, 62.8 δ B. Molecular formula: C7H12O2 Broadband decoupled: 19.1 δ, 28.0 δ, 70.5 δ, 129.0 δ, 129.8 δ, 165.8 δ DEPT-90: 28.0 δ, 129.8 δ DEPT-135 (positive): 19.1 δ, 28.0 δ, 129.8 δ DEPT-135 (negative): 70.5 δ, 129.0 δarrow_forward
- Q2: The proton NMR spectrum is shown for a compound with formula C5H9NO4. The infrared spectrum displays strong bands at 1750 and 1562 cm-1 and a medium-intensity band at 1320 cm-1. The normal carbon- 13 and the DEPT experimental results are tabulated. Draw the structure of this compound. Normal Carbon DEPT-135 DEPT-90 14 ppm Positive No peak No peak 16 Positive 63 Negative No peak 83 Positive Positive 165 No peak No peak Proton spectrum C;H,NO4 0.92 2.01 3.00 3.00 5.0 4.5 4.0 3.5 3.0 25 2.0 1.5 10 0.5 0.0arrow_forward3. Propose a structure for an organic compound with molecular formula C3H1404 given the following 'H NMR and IR spectra. Draw the structure only, no need to interpret the spectra. 1H NMR Spectrum IR Spectrum Triplet Singlet d (6H) 8 (4H) 8 (4H) 1740 cm1 Quartetarrow_forwardQ3: The proton NMR spectrum of a compound with formula C5H100 is shown. The DEPT experimental results are tabulated. The infrared spectrum shows medium-sized bands at 2968, 2937, 2880, 2811, and 2711 cm1 and strong bands at 1728 cm-1. Draw the structure of this compound. Normal Carbon DEPT-135 DEPT-90 11.35 ppm No peak No peak No peak Positive 12.88 Positive 23.55 Negative 47.78 Positive Positive 205.28 Positive Positive (C=0) T m 240 2.35 2.30 2.25 2.20 0.75 0.91 1.04 1.01 2.90 2.92 9.6 9'5 24 23 2.2 2.1 2.0 1.9 1.8 1.7 1.6 14 1.3 1.2 1.1 10 0.9 2890.2 \ 2888.0 690.2 0'889 F189¬ 669.6 667.8arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
