
Concept explainers
Practice Problem F.1
Show how you might use a sulfur ylide to prepare

Interpretation:
The sulfur ylide required for the preparation of the given compounds is to be shown.
Concept introduction:
Sulphur ylide acts as the nucleophile and attacks the carbonyl carbon of aldehydes and ketones.
The intermediate formed in the reaction of sulphur ylide and carbonyl carbon is generally an epoxide rather than an alkene.
Answer to Problem 1PP
Solution:
The reactant used are:
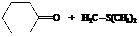
The reactant used are:
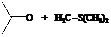
Explanation of Solution
a)
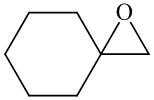
In the formation of the given product, the ketone used is cyclo hexanone. In the first step, sulfur ylide acts as a nucleophile (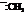 group of sulfur ylide) and attacks the carbonyl carbon of cyclo hexanone. After this,
group of sulfur ylide) and attacks the carbonyl carbon of cyclo hexanone. After this, 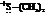 group is removed from the sulfur ylide, which results in the formation of the epoxy ring. The whole reaction can be shown as follows:
group is removed from the sulfur ylide, which results in the formation of the epoxy ring. The whole reaction can be shown as follows:
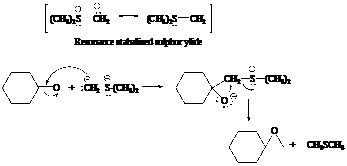
Hence, cyclohexane is used in combination with sulfur ylide to form the given compound.
b)
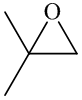
In the formation of the given product, the ketone used is acetone. In the first step, sulfur ylide acts as a nucleophile ( group of sulfur ylide) and attacks the carbonyl carbon of the acetone. After this,
group of sulfur ylide) and attacks the carbonyl carbon of the acetone. After this, 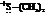 group is removed from the sulfur ylide which results in the formation of the epoxy ring.
group is removed from the sulfur ylide which results in the formation of the epoxy ring.
The whole reaction can be shown as follows:
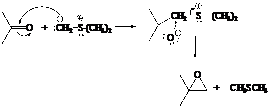
Hence, acetone is used in combination with sulfur ylide to form the given compound.
Want to see more full solutions like this?
Chapter F Solutions
Organic Chemistry
Additional Science Textbook Solutions
Chemistry
Chemistry & Chemical Reactivity
Chemistry: Matter and Change
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
Organic Chemistry (8th Edition)
Living By Chemistry: First Edition Textbook
- What is the IUPAC name of the following substance? ball & stick Submit Answer ŷ + labels Retry Entire Group 9 more group attempts remaining Cengage Learning Cengage Technical Supportarrow_forwardNeed help with providing major productarrow_forwardSelect one of the following: a) I b) II c) III d) IVarrow_forward
- (please show reaekson and incorrect option explain and correct answer)arrow_forwardIndicate whether each of the following compounds is an alcohol, phenol, thiol, or ether using the dropdown on the right. Compound A: Compound B: Compound C: Compound D: Compound E: Compound F: OH ㅅ OH Br OH SHarrow_forwardDetailed explanationarrow_forward
- Consider the following molecular interactions in the liquids (I) H2O --- H2O (II) HF --- HF (III) CH3COCH3 --- CH3COCH3 (IV) C2H5OH --- C2H5OH If the above pairs of molecules are arranged in the order of the strengths of the interactions between them, putting the pair with the strongest interaction first, the order would be. Please type answer note write by hendarrow_forwardPhthalein Dye Test Chemical Reaction Balanced equationarrow_forwardProblem B: Dicholorodiflouromethane, CCl2F2, a known refrigerant, can be produced by the chemical equation shown below. Consider the following: atomic mass of C is 12, Cl is 35, and F is 19. CH4 + Cl2 -> CCl4 + HCl (not balanced) CCl4 + HF -> CCl2F2 + HCl (not balanced) 1. Assuming that all the CCl4 produced in the first reaction is consumed in the second, how many moles of Cl2 must be consumed in the first reaction to produce 2.25kg CCl2F2 in the second? Show the complete solution A. 56.24 mol Cl2 B. 56.25 mol Cl2 C. 56.26 mol Cl2 D. 56.27 mol Cl2 E. 2250g Cl2 F. None of the abovearrow_forward
