
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter F, Problem 4PP
Interpretation Introduction
Interpretation:
The compound British Anti – Lewisite (BAL) is to be synthesised from allyl alcohol.
Concept introduction:
The compound British Anti – Lewisite (BAL) is used as inorganic mercury, which is effective in protecting the kidney from 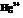 . In BAL, both the thiol groups
. In BAL, both the thiol groups 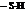 lose hydrogen and binds with the
lose hydrogen and binds with the 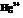 .
.
Addition of bromine across the double bond is done via electrophilic addition reaction. This can be shown as follows:
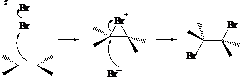
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Provide the correct IUPAC name for VF₂.
hexa-
(1) (1) (1) (IV)
octa-hepta- penta-
tetra- tri- di- mono-
vanadide fluorite fluoride
vanadium fluorine
hydrate acid
Delete
5.21 Give IUPAC names for the following substances (red = 0, blue = N).
(a)
(b)
Identify which of the following compounds are aromatic.
I
II
III
(A) I and II
(B) II and III
(C) Only III
(D) I, II and III
Chapter F Solutions
Organic Chemistry
Knowledge Booster
Similar questions
- 6.2.Q (A) it is the major product of the following Stork reaction? (C) (B) Sue St Stork Reaction (D) ?arrow_forwardAcetic acid is weak acid but it gives strongly acidic solution when dissolved in perchloric acid. Explain.arrow_forwardOptions for both: Compound A, compound B, compound C, compound Darrow_forward
- 7.7 Predict the product of the reaction. HCIarrow_forwardWhich nitrogen atom is the most basic in compound P? H₂N (a) (b) P CN (c) (A) nitrogen a (B) nitrogen b (C) nitrogen c (D) it is impossible to determine the most basic nitrogen atomarrow_forwardProblem 5: Arrartge the following isomeric amines in the order öf increasing boiling point. Provide a succinct rationale A picture or a scheme is worth significantly more than words... NH2 (X) () (Z)arrow_forward
- (b) A compound with the molecular formula C4H9CI has four structural isomers. Write the structural formulas for all the four isomers. (c) Arrange the following compounds in order of increasing acidity, and explain the reasons forarrow_forwardHypoglycin A, an amino acid derivative found in unripened lychee, is anacutely toxic compound that produces seizures, coma, and sometimesdeath in undernourished children when ingested on an empty stomach. (a) Draw the neutral, positively charged, and negatively charged forms of hypoglycin A. (b) Which form predominates at pH = 1, 6, and 11? (c) What is the structure of hypoclycin A at its isoelectric point?arrow_forwardWhich of the following compounds exhibit hydrogen bonding in the pure liquid state? (The lone pair electrons are NOT shown) (A) hydrogen peroxide, formaldehyde, formic acid, and 1, 1-difluoroethane (B) hydrogen peroxide and formic acid (C) hydrogen peroxide, formaldehyde, and formic acid (D) formaldehyde, formic acid, and difluoroethane (E) hydrogen peroxide, formic acid, and 1, 1-difluoroethanearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY