
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter D, Problem 2PP
Practice Problem D.2
(a) Show the orbitals involved in the following thermal electrocyclic reaction.
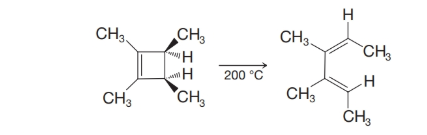
(b) Do the groups rotate in a conrotatory or disrotatory manner?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
PROBLEM F.6 Draw the structure for each of the following molecules.
2,2-dimethylcyclopentanecarboxylic acid; (b) cyclohepta-3,5-diene-1-carboxamide;
acid;
(a)
(c) (1R, 4S)-4-cyanocyclooctane-1-carboxylic
(d) (15,2S)-2-hydroxy-N-(1-methylethyl)cyclobutane-1-carboxamide;
(e) (R)-cyclohexa-2,4-diene-1-carbonitrile
Concept Check No. 4 (Continued). For Problem No. 7.29 (q) to (v),
IDENTIFY the given pair of structures as described below and
WRITE:
EN if the pair represents ENANTIOMERS
DI if the pair represents DIASTEREOMERS
Co if the pair represents CONSTITUTIONAL isomers
ID if the two molecules represent IDENTICAL compounds
(q) H,C-
OH
and
H,C-
OH
(r) (CH,),C
and
(CH,),C-
(s)
and
-CH3
CH3
H3C
(t)
and
CH,
H3C.
H.
CH,
(u)
and
H,C.
H.
CH3
(v)
and
Answers:
(q)
(r)
(s)
(t)
(u)
(v)
Give clear explanation answer
Chapter D Solutions
Organic Chemistry
Ch. D - Prob. 1PPCh. D - Practice Problem D.2 (a) Show the orbitals...Ch. D - Prob. 3PPCh. D - Prob. 4PPCh. D - Prob. 5PPCh. D - Prob. 6PPCh. D - Practice Problem D.7 Can you suggest a...Ch. D - Practice Problem D.8 When compound A is heated,...Ch. D - Prob. 9PPCh. D - Practice Problem D.10 What reactant could lead to...
Additional Science Textbook Solutions
Find more solutions based on key concepts
10.1 Indicate whether each of the following statements is characteristic of an acid, a base, or
both:
has a so...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Look up the physical properties of several of the compounds you will be making in your lab from several differe...
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
1. What are the standard SI base units of length, mass, time, and temperature?
Chemistry: Structure and Properties (2nd Edition)
Compound T (C5H8O) has a strong IR absorption band at 1745 cm1. The broad-band proton decoupled 13C spectrum of...
Organic Chemistry
1.6 Read the labels on products used to wash your dishes. What are the names of some chemicals contained in tho...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
What is the pHofa0.20M solution of Na2SO3at25C? For the diprotic acid, H2SO3,Ka1=1.510-2andKa2=6.310-8.
Chemistry: The Molecular Nature of Matter
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Problem Carrow_forwardWhat is each compounds systematic name? please answer both compoundarrow_forwardThe degree of unsaturation, or index of hydrogen deficiency, is the number of pi bonds plus rings in a molecule. Specify the degree of unsaturation (index of hydrogen deficiency) of the following formulas: (a) C18H18 (b) CsHgO (c) C10H8N₂ Try Another Version 3 item attempts remaining Submit Answerarrow_forward
- The green pathway: the electrons flow from the negatively charged oxygen atom to the C-O bond, the C=c bond to the C-C bond, the C=C bond to the carbon atom, and the carbon atom to the hydrogen atom than the oxygen atom (A) I dont understand why is the proton transfer from the hydrogen atom to the oxygen atom needed (B) I do not understand why cant the electron flow directly from the O - to the oxygen atom in H2O?arrow_forwardProvide the correct IUPAC name for VF₂. hexa- (1) (1) (1) (IV) octa-hepta- penta- tetra- tri- di- mono- vanadide fluorite fluoride vanadium fluorine hydrate acid Deletearrow_forwardProblem 2.2 For each compound below, identify the atom most likely to be attacked first by a nucleophile and identify the unoccupied frontier orbital (for example p, T", or σ*) associated with that atom. Clarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
CBSE Class 12 Chemistry || Polymers || Full Chapter || By Shiksha House; Author: Best for NEET;https://www.youtube.com/watch?v=OxdJlS0xZ0Y;License: Standard YouTube License, CC-BY