
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter D, Problem 12PP
Practice Problem D.12
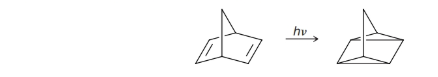
Show what happens in the following reactions:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
answer IS
Predict the major product of the following process.
1) LDA
i
?
2)
3) H3O+
17.27 Predict the product for each of the following reactions:
(a)
(b)
Heat
Heat
?
?
?
(c)
lonete
Heat
?
loosbyris
(d)
?
Heat be
ent
(a)
H.
H.
6.44 Which of the following reactions will yield a racemic
mixture of products?
HCI
HBr
(a)
(c)
H2
H2
(b)
(d)
Pt
Pt
Chapter D Solutions
Organic Chemistry
Ch. D - Prob. 1PPCh. D - Practice Problem D.2 (a) Show the orbitals...Ch. D - Prob. 3PPCh. D - Prob. 4PPCh. D - Prob. 5PPCh. D - Prob. 6PPCh. D - Practice Problem D.7 Can you suggest a...Ch. D - Practice Problem D.8 When compound A is heated,...Ch. D - Prob. 9PPCh. D - Practice Problem D.10 What reactant could lead to...
Additional Science Textbook Solutions
Find more solutions based on key concepts
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
Basic Chemistry (5th Edition)
When 20g of aluminum and 20 g of copper at 200C are placed into ice water, the one that cools faster needs to b...
Living by Chemistry
The reaction of tert-butyl chloride with methanol (CH3)3CCl + CH3OH (CH3)3COCH3 + HCl tert butyl chloride met...
Organic Chemistry (9th Edition)
1.3 Obtain a bottle of multivitamins and read the list of ingredients. What are four chemicals from the list?
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
27. Consider the reaction.
Express the rate of the reaction in terms of the change in concentration of each of...
Chemistry: Structure and Properties (2nd Edition)
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Select all possible products of the following questionarrow_forwardExplain why the reaction in Problem 18.48 proceeds dramatically more slowly under neutral conditions than under either acidic or basic conditions.arrow_forwardWhat compound undergoes metathesis to form each of the following compounds?arrow_forward
- By using only electron density arguments, determine whether the following reactions will occur.arrow_forwardHave this practice synthesis problem in my textbook. I think the final solution might include the formation of a cyclic compound. Any assistance in understanding the solution to this problem would be greatly appreciated. Thanks!arrow_forwardPredict the product for the given reaction. A A B monit NONE OF THE ABOVE B heat Carrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Introduction to Organometallic Compounds; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=3FRV31YYtL8;License: Standard YouTube License, CC-BY