
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter D, Problem 13PP
Practice Problem D.13
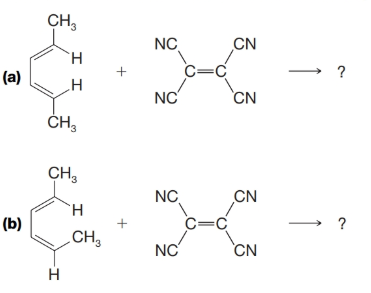
What products would you expect from the following reactions?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Predict the major product of the following reactions. The formulae of the products are given in bold.
Please answer d,e,f,g
874
Problem 7 of 20
Submit
Draw the product of the reaction shown
below. Ignore inorganic byproducts.
N
+
H3C CH3
Br
Q
Sn
CH3
Pd(dba)2, PPh3, CSF
dioxane
Select to Draw
Q
Problem 2,8
Suggest a plausible arrow-pushing mechanism for the following tautomerization
reactions
cat. HA
OH
cat B
O NH2
NH2
NIH
Chapter D Solutions
Organic Chemistry
Ch. D - Prob. 1PPCh. D - Practice Problem D.2 (a) Show the orbitals...Ch. D - Prob. 3PPCh. D - Prob. 4PPCh. D - Prob. 5PPCh. D - Prob. 6PPCh. D - Practice Problem D.7 Can you suggest a...Ch. D - Practice Problem D.8 When compound A is heated,...Ch. D - Prob. 9PPCh. D - Practice Problem D.10 What reactant could lead to...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
1.1 Write a one-sentence definition for each of the following:
a. chemistry
b. chemical
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Explain why 92% of 2,4-pemtanedione exists as the enol tautomer in hexane but only 15% of this compound exists ...
Organic Chemistry
The oxidation of sulfur dioxide to give sulfur trioxide is an important step in the industrial process for the ...
CHEMISTRY-TEXT
Whether change in phase is a physical or a chemical change needs to be determined. Concept introduction: The na...
Chemistry: Matter and Change
A mixture of gases A2 and B2 are introduced to a slender metal cylinder that has one end closed and the other ...
Chemistry: The Central Science (13th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What reagents are required to carry out the following reaction (select all that apply)? a HNO3 b H2SO4 c Cl2 d Br2 e H2O f CH3COCl g AlCl3 h FeCl3 i FeBr3 j CH3CHClCH3arrow_forwardPredict the product for the following sigmatropic reactions. CH3 Practice Problem C₂H5 CH3 [3,3]-C Enolisation [5,5]-C Enolisationarrow_forwardIdentify the products of the following reaction. A) H 1) MCPBA 2) H3O+ H3C H3C HO OH H CH3 HO OH B) H&C H H3C CH3 H3C H3C HO OH H CH3 OH D) H3C H H3C CH3 (Select all that apply) U Aarrow_forward
- Synthesis practice ork assign 1) Synthesize this compound only using only organic starting materials such as acetic anhydride, triphenylphosphine, ethyne, and alcohols of four or fewer carbons and any inorganic reagents needed CH3 I CH3CH₂-C-C=CH I ОНarrow_forwardProblem 18 of 50 Submit Curved arrows are used to illustrate the flow of electrons. Follow the arrows and draw the product formed in this reaction. Include all lone-pairs. Ignore any inorganic byproducts. HNO3 H2SO4 مل علم Нarrow_forwardWhat is the expected major product of the following reaction (select all that apply)? SeH Br2 FeBr SeH Br SeH A Br B Br SeH SeBr с Darrow_forward
- nt Paragraph Predict the main product of each of the following reactions: CH3CH2OH (a) 1-bromopropane + NaOCH2CH3 CH3OH (b) 2-bromo-2-methylbutane + NaOCH3 127 |arrow_forwardIdentify the major product(s) for the following reaction. Select all that apply. If the reaction does not yield a product, select "No reaction". hv No reactionarrow_forward23) Which of the following reactions will not produce the given product in good yield? Select all that apply Reagent only reacts once MeONa a) Br MEOH Br Br (CH3),CONa b) Br (CH3)3COH H2N OH 1)(CH3)3CONa 2)arrow_forward
- Problem Three a) Identify the following as nucleophile or electrophile: iii) C,H,CHO vi) HOC₂H₂ i) C,H5NH, iv) (C$Hs)₂CO ii) (C₂Hs)3N v) (C₂H5)₂CH+ viii) CH,SH vii) CN ix) CH₂CH₂CH₂CH₂ Br b) i) Draw the curly arrow mechanism for the reaction between v) and vii) x) (CH3)3CBr ii) of the two C atoms involved do any change their hybridisation? If so, how?arrow_forwardQuestion 14 Unknown X, C5H9BT does not react with bromine or with dilute KMN04. Upon treatment with potassium tert-butoxide, X gives only one product, Y, C5H8 Unlike X, Y decolorizes bromine and changes KMNO4 from purple to brown. Catalytic hydrogenation of Y gives methylcyclobutane. Ozonolysis-reduction of Y gives dialdehyde Z C5H802 Propose corisistent structures for X, Y, and Z. Is there any aspect of the structure of X that is still unknown?arrow_forwardProblem 29 of 50 Submit Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic steps. Be sure to account for all bond- breaking and bond-making steps. N Select to Add Arrows THF Na Ⓒ Na O Oarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY