
Interpretation:
The product of photochemical cyclization reaction of
Concept Introduction:
>The compounds that have the same molecular formula of atoms but the structure of the compounds are different for the positions of atoms are called isomers.
>In double bond or cyclic compounds, if two same functional groups are present on the same side of the double bond or cyclic compound, the given compound can be labeled as cis.
>If the two functional groups are present on the different sides of the double bond or cyclic compound, the given compound can be labeled as Trans.
>Cis-trans isomerism exists in the compounds in which similar groups are present on the adjacent carbon atoms.
>The thermal electrocyclic reaction having
The photochemical electrocyclic reaction having
The electrocyclic reactions are initiated by heat or light and are stereospecific in nature.
Answer to Problem 1PP
Solution:
The product of the reaction is given below:
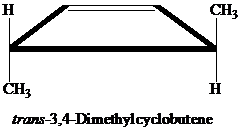
Explanation of Solution
The compound
Thus, the photochemical cyclization reaction of

The product of the reaction is
Want to see more full solutions like this?
Chapter D Solutions
Organic Chemistry
- Hydrolysis of 3-fluoro-3-methylhexane will most likely follows_______ mechanism. A. SN1 B. E1 C. SN2 D. E2arrow_forwardFor each reaction below, provide the product(s) that would form and indicate whether the substitution reaction would occur by an SN1 , SN2, E1, or E2 mechanism. Draw all constitutional and/or stereoisomers that would form in each reaction. I need help with this practice problem, thank you!arrow_forwardThe hydrolysis of CH3-CH=CH-CH2-Cl gives rise to two products: CH3-CH=CH-CH2-OHl and CH3-CH(-OH)CH=CH2. Write a mechanism that accounts for their formation. Show each step clearly. Use curved arrow notation.arrow_forward
- Mechanism questionarrow_forwardWhen cyclohexanone reacts with 2 moles of ethanol and HCl catalyst, cyclohexanone diethyl ketal is produced. Draw a plausible mechanismarrow_forwardtrue or false question. ( explanation not needed) a) Gilman copper reagent does 1,2 nucleophilic addition to α,β.unsaturated ketones b)Tertiary alcohol will be formed by the reaction between any ketone and Grignard. c)Cyclopentadiene anion is aromatic d)Tertiary alcohols undergoes dehydration by E1 mechanism e)1,4-addition product is the kinetic product in the addition of conjugated diene f) Benzene is more reactive than toluene in aromatic electrophilic substitution reactionarrow_forward
- List carbon-carbon bond formation reactions to synthesize 1-bromohexane from starting materials with no more than three carbon atoms. Please draw out processarrow_forwardWhat is the correct stereochemistry of the nucleophilic addition product(s) of the reactants shown below? Select all that are correct.arrow_forwardMechanism Aniline from nitrobenzenearrow_forward
- Problem 2: Consider the following reaction. Using mechanism and less than 30 words explain the outcome of this reaction.arrow_forwardShow synthetic strategy to convert 1-bromo-1-methycyclohexane into 1-(2-methylpropyl)cyclohexene. Neither mechanism nor stereochemistry are required for this question.arrow_forwardQuestion: Draw structures for the thermodynamic products of the following reaction. Pay careful attention to stereochemistry. **Can you assist me in idenifying why the models are incorrect? Any feedback would be greatly appreciated. Thank you.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





