
Concept explainers
If lactic acid is similar in strength to acetic acid (Table. 9-2), predict whether reactants or products are favored in each reaction. 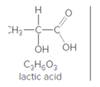
a.
lactic acid
b.
lactic acid
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Table 13-4 lists the stepwise Ka values for some polyprotic acids. What is the difference between a monoprotic acid, a diprotic acid, and a triprotic acid? Most polyprotic acids are weak acids; the major exception is H2SO4. To solve for the pH of a solution of H2SO4, you must generally solve a strong acid problem as well as a weak acid problem. Explain. Write out the reactions that refer to Ka1 and Ka2 for H2SO4. For H3PO4, Ka1 = 7.5 103, Ka2 = 6.2 108, and Ka3= 4.8 1013. Write out the reactions that refer to the Ka1, Ka2and Ka3equilibrium constants. What are the three acids in a solution of H3PO4? Which acid is strongest? What are the three conjugate bases in a solution of H3PO4? Which conjugate base is strongest? Summarize the strategy for calculating the pH of a polyprotic acid in water.arrow_forwardWhat is the ionization constant at 25 C for the weak acid CH3 NH3+, the conjugate acid of the weak base CH3NH2, Kb=4.4104.arrow_forwardWhich ligand is most likely to stabilize a metal in a low oxidation state? O H2O NO O NH3arrow_forward
- If AG° < 0, then at equilibrium there will be more products than reactants. false truearrow_forwardFor each of the following reactions, tell whether the equilibrium will favor the reactants or the products. Use the Ka values in the picture. a. HNCO(aq) + CN– (aq) ⇄⇄ NCO– (aq) + HCN (aq) b. HCO2H (aq) + OH– (aq) ⇄⇄ CO2H– (aq) + H2O (l)arrow_forwardWhat is the approximate K for the following acid/base reaction? H25 (aq) +F (aq) ++ HF (aq) +HS (aq) ACID BASE HCI C H2SO4 HNO3 H30 (aq) HSO H3PO4 HSO NO3 H2O so, H,PO, F HF CH3COOH H,CO3 H,S H,PO, NH, CH;COO HCO, HS HPO, NH, CO PO OH HCO HPO. 1,0 OH H, CH CH, OA unable to determine OB. K.<1 Base strength increasing Acid strength increasingarrow_forward
- For 6 and 7, use the table of K, values compiled at 25 °C to answer the questions below: Acid HNO3 HCI HBr H2SO4 HSO41 HF HNO2 HCOOH Infinite Infinite Infinite Infinite Ka 1.3 x 102 7.1 x 104 4.5 x 104 1.7 x 104 (Strong acid) HC2041 6.1 x 105 (Strong acid) CH3COOH (Strong acid) (Strong acid) HCO31 4.8 x 1011 Acid C6H5COOH H2CO3 H2PO41 HCN НРОД? Ka 6.5 x 105 1.8 x 106 4.2 x 107 6.2 x 108 4.9 x 1010 4.8 x 1013 6. Which of the following has the highest pH? a. 1.00 M NaNO3 b. 1.00 M NaNO2 с. 1.00 М NaH2РОд d. All have the same pH 7. Which of the following has the lowest pH in aqueous solution? a. 1.00 M NaCl b. 1.00 M NaNO3 c. 1.00 M NaBr d. All have the same pHarrow_forwardThe dissociation of 0.15 M phosphoric acid is given by:H3PO4(aq) + H2O(l) ⇄ H2PO4^-(aq) + H3O^+(aq) Ka = 7.1 x 10-3 (at 25°C) H2PO4^-(aq) + H2O(l) ⇄ HPO4^2-(aq) + H3O^+(aq) Ka = 6.3 x 10-8 (at 25°C) HPO4^2-(aq) + H2O(l) ⇄ PO4^3-(aq) + H3O^+(aq) Ka = 6.3 x 10-8 (at 25°C) A. Classify the species, H3PO4, H2PO4-, HPO4^2-, PO4^3-, as acid, base, or ampholyte.B. Applying the systematic method in solving acid-base equilibria problems, calculate the pH of the following:a. 0.050 M H3PO4 b. 0.050 M NaH2PO4 c. 0.050 M Na2HPO4d. 0.050 M Na3PO4arrow_forwardThe dissociation of 0.15 M phosphoric acid is given by: H3PO4(aq) + H2O(l) ⇄ H2PO4^-(aq) + H3O^+(aq) Ka = 7.1 x 10-3 (at 25°C) H2PO4^-(aq) + H2O(l) ⇄ HPO4^2-(aq) + H3O^+(aq) Ka = 6.3 x 10-8 (at 25°C) HPO4^2-(aq) + H2O(l) ⇄ PO4^3-(aq) + H3O^+(aq) Ka = 6.3 x 10-8 (at 25°C) B. Applying the systematic method in solving acid-base equilibria problems, calculate the pH of the following: a. 0.050 M H3PO4 b. 0.050 M NaH2PO4 c. 0.050 M Na2HPO4 d. 0.050 M Na3PO4arrow_forward
- When propionic acid is reacted with sodium hydroxide, as shown below, what can be said about the reaction after equilibrium has been reached? HO, NaOH + H,0 + Na propionic acid sodium hydroxide sodium propionate NaOH = Na® OH %3D O the concentration of propionic acid and sodium propionate are approximately equal the concentration sodium propionate is much greater than the concentration of propionic acid the concentration of propionic acid is much greater than the concentration of sodium propionatearrow_forwardConsider Le Châtelier’s Principle. In which direction does the addition of a competitiveinhibitor shift the original E + S ⇌ ES equilibrium?arrow_forwardConsider the following acid-base reaction: H3O+ + HPO42 H₂PO4 + H₂O Identify the Bronsted-Lowry base. OH₂O ⒸHPO4 ²- OH₂PO4 OH3O+ What is the concentration (in Molarity) of a Sulfuric Acid (H₂SO4) solution if a 15.0 mL sample required 26.4 mL of 0.950 M KOH for neutralization? H₂SO4 + 2 KOH --> K₂SO4 + 2 H₂O (balanced) 0.012 M 0.025 M 0.836 M 0.270 Marrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





