
Concept explainers
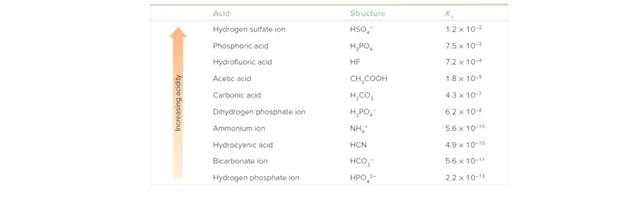
Label the acid in the reactants and the conjugate acid in the products in each reaction. Use the data in Tables 9.2 and 9.3 to determine whether the reactants or products are favored at equilibrium. Explain your reasoning.
a.
b.
c.
Table 9.2 Relative Strength of Acids and Their Conjugate Bases
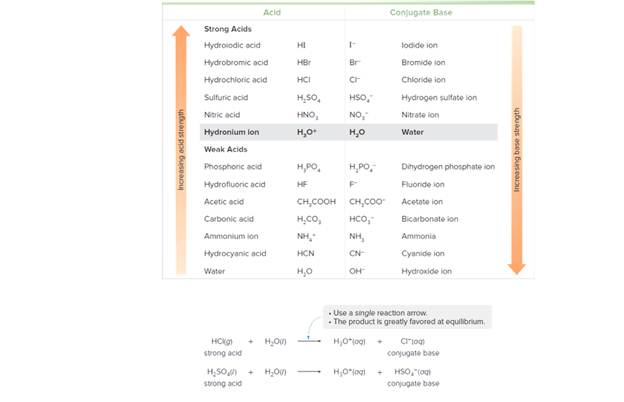 Table 9.3 Acid Dissociation Constants
Table 9.3 Acid Dissociation Constants
Trending nowThis is a popular solution!

Chapter 9 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Most naturally occurring acids are weak acids. Lactic acid is one example. CH3CH(OH)CO2H(s)+H2O(l)H3O+(aq)+CH3CH(OH)CO2(aq) If you place some lactic acid in water, it will ionize to a small extent, and an equilibrium will be established. Suggest some experiments to prow that this is a weak acid and that the establishment of equilibrium is a reversible process.arrow_forwardPredict which of these acid-base reactions are product-favored and which are reactant-favored. In each case write a balanced equation for any reaction that might occur, even if the reaction is reactant-favored. Consult Table 14.2 if necessary. H2O(ℓ) + HNO3(aq) H3PO4(aq) + H2O(ℓ) CN−(aq) + HCl(aq)arrow_forwardPredict which of these acid-base reactions are product-favored and which are reactant-favored. In each case write a balanced equation for any reaction that might occur, even if the reaction is reactant-favored. Consult Table 14.2 if necessary. (a) NH4+(aq)+HPO42(aq) (b) CH3COOH(aq) + OH(aq) (c) HSO4(aq)+H2PO4(aq) (d) CH3COOH(aq) + F(aq)arrow_forward
- Consider the following acidic equilibrium: H₂CO₃(aq) + H₂O(l) ⇌ HCO₃⁻(aq) + H₃O⁺(aq). If you add NaHCO₃ to this solution, which of the following will occur? A) The reaction quotient will decrease. B) The reaction will shift in the reverse direction. C) The equilibrium constant will increase. D) No changes to the equilibrium positions will take place.arrow_forwardCH3CH2COOH Draw the Lewis structure of the acid and mark the acidic hydrogen with an asterisk (*). Draw a Lewis structure of the conjugate base of the acid. Suppose the acid is neutralized with a strong base. a) Which of the two structures you drew in 6 and 7 would be the predominant form of the species at a pH well above that at the equivalence point? b) Which of the two structures would be the predominant form of the species at a very low pH, well below that at the equivalence point and similar to the pH near the start of the titration? c) At what point in the titration, if any, would there be equal amounts of the two forms?arrow_forward1. The odor of spoiled butter is due in part to butanoic acid (HC4H7O2) which results from the breakdown of the fat in butter. A 0.100M solution of butanoic acid is 1.23% ionized. a. Write the chemical equation for the ionization of this acid in water. b. Write the Ka expression for this acid. c. What is the equilibrium concentration of each product and the reactant?arrow_forward
- The pH of a solution containing a hydrogen ion concentration of 1 x 10-5 is: A. 3 and its pOH is 6B. 3 and its pOH is 7C. 5 and its pOH is 8D. 5 and its pOH is 9arrow_forwardWhat is the approximate K for the following acid/base reaction? H25 (aq) +F (aq) ++ HF (aq) +HS (aq) ACID BASE HCI C H2SO4 HNO3 H30 (aq) HSO H3PO4 HSO NO3 H2O so, H,PO, F HF CH3COOH H,CO3 H,S H,PO, NH, CH;COO HCO, HS HPO, NH, CO PO OH HCO HPO. 1,0 OH H, CH CH, OA unable to determine OB. K.<1 Base strength increasing Acid strength increasingarrow_forwardConsider the following equilibrium: HNO2(aq) + H20 (1) = H3O*(aq) + NO2 (aq) In which direction (if any) will the equilibrium shift if: i. KNO2 is added? i. NaOH is added?arrow_forward
- For each of the following reactions, identify the acid and the base. Also indicate which acid- base definition (Lewis, Brønsted-Lowry) applies. In some cases, more than one definition may apply. a. AlBr3 + Br → AIBr, b. HCIO4 + CH;CN→ CH;CNH† + CIO4¯ c. Ni2+ + 6 NH3 d. NH3 + CIF→H¿N•CIF →[Ni(NH3)6]** -arrow_forwardComplete the reaction by writing the formulas of the products. CH;COOH + NH, = The Ką of CH, COOH (acetic acid) is 1.8 × 10-5. The Ką of the conjugate acid formed in this reaction is 5.6 x 10-10. Indicate whether the products or the reactants are favored. The reactants are favored. The products are favored.arrow_forwardHeroin, a derivative of morphine, is a powerful analgesic and a powerful narcotic agent. Calculate K, for heroin if the pH of a 1.7 x 10-3 M solution was found to be 9.60. O 9.5 x 10-7 2.3 x 10-2 O 9.3 x 10-7 O 1.5 x 10-7arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning





