
The exergy destruction associated with each process of the Brayton cycle and the exergy of the exhaust gases at the exit of the regenerator.
Answer to Problem 144P
The exergy destruction associated with process 1-2 of the given Brayton cycle is
The exergy destruction associated with process 3-4 of the given Brayton cycle is
The exergy destruction associated with regeneration process of the given Brayton cycle is
The exergy destruction associated with process 5-3 of the given Brayton cycle is
The exergy destruction associated with process 6-1 of the given Brayton cycle is
The exergy of the exhaust gases at the exit of the regenerator is
Explanation of Solution
Show the regenerative Brayton cycle with air as the working fluid, on
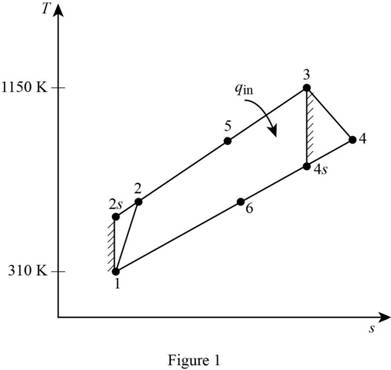
For the given Brayton cycle with regeneration and air as the working fluid
Write the pressure and relative pressure relation for the process 1-2.
Write the pressure and relative pressure relation for the process 3-4.
Write the expression of efficiency of the compressor
Write the expression of efficiency of the turbine
Write the expression of net work output by the gas turbine
Here, work done by the turbine is
Write the expression of effectiveness of the regenerator
Write the expression of heat input to the regenerative Brayton cycle
Write the expression of heat rejected by the regenerative Brayton cycle
Write the expression of thermal efficiency of the given turbine
Write the energy balance equation on the heat exchanger.
Write the expression of exergy destruction associated with the process 1-2 of the given Brayton cycle
Here, the temperature of the surroundings is
Write the expression of exergy destruction associated with the process 3-4 of the given Brayton cycle
Here, entropy of air at state 3 as a function of temperature is
Write the expression of exergy destruction associated with the regeneration process of the given Brayton cycle
Here, entropy of air at state 5 as a function of temperature alone is
Write the expression of exergy destruction associated with the process 5-3 of the given Brayton cycle
Here, the temperature of the heat source is
Write the expression of exergy destruction associated with the process 6-1 of the given Brayton cycle
Here, the temperature of the sink is
Write the expression of stream exergy at the exit of the regenerator (state 6)
Here, the specific enthalpy of the surroundings is
Write the expression of change entropy for the exit of the regenerator
Here, entropy of air at the surroundings as a function of temperature alone is
Conclusion:
Refer Table A-17, “Ideal gas properties of air”, obtain the properties of air at a temperature of
Substitute 7 for
Refer Table A-17, “Ideal gas properties of air”, obtain the properties of air at a relative pressure of 10.88
Refer Table A-17, “Ideal gas properties of air”, obtain the properties of air at a temperature of
Substitute
Refer Table A-17, “Ideal gas properties of air”, obtain the properties of air at a relative pressure of 28.59
Rearrange Equation (III), and substitute
Refer Table A-17, “Ideal gas properties of air”, obtain the properties of air at a enthalpy of
Rearrange Equation (IV), and substitute
Refer Table A-17, “Ideal gas properties of air”, obtain the properties of air at a enthalpy of
Substitute
Substitute 0.65 for
Refer Table A-17, “Ideal gas properties of air”, obtain the properties of air at a enthalpy of
Substitute
Substitute
Substitute
Substitute
Refer Table A-17, “Ideal gas properties of air”, obtain the properties of air at a enthalpy of
Substitute 290 K for
Thus, the exergy destruction associated with process 1-2 of the given Brayton cycle is
Substitute 290 K for
Thus, the exergy destruction associated with process 3-4 of the given Brayton cycle is
Substitute 290 K for
Thus, the exergy destruction associated with regeneration process of the given Brayton cycle is
Substitute 290 K for
Thus, the exergy destruction associated with process 5-3 of the given Brayton cycle is
Substitute 290 K for
Thus, the exergy destruction associated with process 6-1 of the given Brayton cycle is
Refer Table A-17, “Ideal gas properties of air”, obtain the properties of air at a temperature of
Substitute
Substitute
Thus, the exergy of the exhaust gases at the exit of the regenerator is
Want to see more full solutions like this?
Chapter 9 Solutions
Thermodynamics: An Engineering Approach
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





