
Organic Chemistry (8th Edition)
8th Edition
ISBN: 9780134042282
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 83P
Draw the major product obtained when each of the following
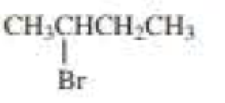
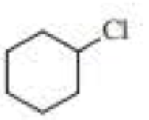
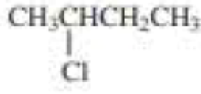
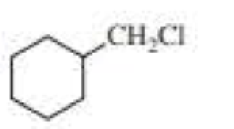
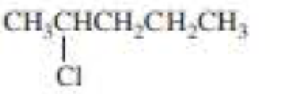
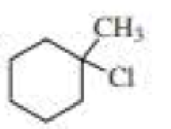
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Part A
What is the major product formed when the following compound undergo an E1 reaction?
CH, CH
CH;CH,C–CHCH;CH3
Br
for bonds ond chargee) Atoms gnd Ter
What is the major product formed when the following compounds undergo an E1 reaction?
What products are obtained from the reaction of each of the following alkenes with OsO4 followed by aqueous H2O2?
Chapter 9 Solutions
Organic Chemistry (8th Edition)
Ch. 9.1 - Prob. 3PCh. 9.1 - Does increasing the energy barrier for an SN2...Ch. 9.1 - Rank the following alkyl bromides from most...Ch. 9.1 - Prob. 8PCh. 9.2 - Prob. 9PCh. 9.2 - Prob. 10PCh. 9.2 - Prob. 11PCh. 9.2 - Prob. 12PCh. 9.2 - Which substitution reaction lakes place more...Ch. 9.2 - Prob. 15P
Ch. 9.2 - Prob. 17PCh. 9.3 - Prob. 18PCh. 9.4 - Prob. 19PCh. 9.5 - Draw the configuration(s) of the substitution...Ch. 9.5 - Which of the following reactions take place more...Ch. 9.7 - Prob. 22PCh. 9.7 - Prob. 23PCh. 9.7 - Prob. 24PCh. 9.7 - Prob. 25PCh. 9.8 - Four alkenes are formed from the E1 reaction of...Ch. 9.8 - If 2-fluoropentane could undergo an E1 reaction,...Ch. 9.8 - Prob. 28PCh. 9.8 - Propose a mechanism for the following reaction:Ch. 9.9 - Prob. 30PCh. 9.9 - What percentage of the reaction described in...Ch. 9.10 - Prob. 33PCh. 9.10 - Prob. 35PCh. 9.11 - Why do cis-1-bromo-2-ethylcyclohexane and...Ch. 9.11 - Which isomer reacts more rapidly in an E2...Ch. 9.11 - Prob. 38PCh. 9.12 - Prob. 39PCh. 9.12 - Prob. 40PCh. 9.12 - Prob. 41PCh. 9.12 - Explain why only a substitution product and no...Ch. 9.12 - a. Explain why 1-bromo-2,2-dimethylpropane has...Ch. 9.13 - Prob. 44PCh. 9.13 - Prob. 45PCh. 9.13 - What products will be obtained from the El...Ch. 9.13 - Prob. 47PCh. 9.13 - Prob. 48PCh. 9.13 - Prob. 49PCh. 9.13 - Why is a cumulated diene not formed in the...Ch. 9.13 - What product is obtained when the following...Ch. 9.13 - What products are formed from the following...Ch. 9.14 - Prob. 54PCh. 9.14 - Prob. 55PCh. 9.14 - Prob. 56PCh. 9.14 - Prob. 58PCh. 9.14 - Under which of the following reaction conditions...Ch. 9.15 - A small amount of another organic product is...Ch. 9.15 - What is the best way to prepare the following...Ch. 9.15 - Prob. 62PCh. 9.15 - What products (including stereoisomers, if...Ch. 9.16 - After a proton is removed from the OH group, which...Ch. 9.16 - Prob. 65PCh. 9.17 - Prob. 66PCh. 9 - Prob. 1PCh. 9 - Methoxychlor is an insecticide that was intended...Ch. 9 - Prob. 67PCh. 9 - Prob. 68PCh. 9 - Prob. 69PCh. 9 - Prob. 70PCh. 9 - Prob. 71PCh. 9 - Prob. 72PCh. 9 - Starting with cyclohexene, how can the following...Ch. 9 - Prob. 74PCh. 9 - The pKa of acetic acid in water is 4.76. What...Ch. 9 - Prob. 76PCh. 9 - Prob. 77PCh. 9 - Prob. 78PCh. 9 - Prob. 79PCh. 9 - Prob. 80PCh. 9 - Prob. 81PCh. 9 - Prob. 82PCh. 9 - Draw the major product obtained when each of the...Ch. 9 - Prob. 84PCh. 9 - a. Indicate how each of the following factors...Ch. 9 - Prob. 86PCh. 9 - A chemist wanted to synthesize the...Ch. 9 - Prob. 88PCh. 9 - Prob. 89PCh. 9 - Prob. 90PCh. 9 - Prob. 91PCh. 9 - Starting with an alkyl halide, how could the...Ch. 9 - Indicate which species in each pair gives a higher...Ch. 9 - Prob. 94PCh. 9 - Rank the following from most reactive to least...Ch. 9 - For each of the following alkyl halides, indicate...Ch. 9 - Prob. 97PCh. 9 - When 2-bromo-2,3-dimethylbutane reacts with a...Ch. 9 - Prob. 100PCh. 9 - When the following compound undergoes solvolysis...Ch. 9 - cis-1-Bromo-4-tert-butylcyclohexane and...Ch. 9 - Draw the substitution and elimination products.Ch. 9 - tert-Butyl chloride undergoes solvolysis in both...Ch. 9 - Prob. 105PCh. 9 - Prob. 106PCh. 9 - In which solventethanol or diethyl etherwould the...Ch. 9 - Prob. 108PCh. 9 - Two bromoethers are obtained from the reaction of...Ch. 9 - Prob. 110PCh. 9 - Prob. 111PCh. 9 - Prob. 112PCh. 9 - Which of the following hexachlorocyclohexanes is...Ch. 9 - Explain why the rate of the reaction of...Ch. 9 - Prob. 115PCh. 9 - Two elimination products are obtained from the...Ch. 9 - Draw the structures or the product of the obtained...Ch. 9 - How could you prepare the following compounds from...Ch. 9 - cis-4-Bromocyclohexanol and...Ch. 9 - Prob. 120PCh. 9 - Propose a mechanism for the following reaction:Ch. 9 - Prob. 122PCh. 9 - Prob. 123PCh. 9 - Prob. 124PCh. 9 - Prob. 125PCh. 9 - Predict the product for the following reaction and...Ch. 9 - Prob. 127PCh. 9 - Prob. 128PCh. 9 - When equivalent amounts of methyl bromide nod...Ch. 9 - Prob. 130PCh. 9 - The reaction of chloromethane with hydroxide ion...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the major product formed when the following compound undergoes an E1 reaction?arrow_forwardRank the following carbonyl compounds in order of increasing reactivity in nucleophilic substitution reactions: H I II III IV ZIarrow_forwardDraw the major organic product formed in each of the following transition-metal catalyzed reactions:arrow_forward
- Which of the following compounds will not undergo keto-enol tautomerism? ОН OH ОН OH ОНarrow_forwardDraw the starting structure that would lead to the major product shown under the provided conditions. Drawing H3O+ HOarrow_forwardDraw the structures of the initially formed enol tautomers in the reactions of propyne and dicyclohexylethyne with dicyclohexylborane followed by NaOHNaOH and H2O2H2O2arrow_forward
- Which of the following sequences of reactions would convert toluene to 2-bromo-4-cyanotoluene? ? Br CN Nitration, bromination, reduction, diazotization, reaction with cyanide anion Bromination, nitration, reduction, diazotization, reaction with cyanide anion Bromination, nitration, diazotization, reduction , reaction with cyanide anion Nitration, bromination, diazotization, reduction, reaction with cyanide anionarrow_forwardDetermine a stepwise mechanism for the following reaction that illustrates why two substitution products are formed. Explain why 1-bromohex-2-ene reacts rapidly with a weak nucleophile (CH3OH) under Syl reaction conditions, even though it is a 1° alkyl halide. 1-bromohex-2-ene Part 1: Br CH₂OH The first step in the reaction proceeds according to which mechanism? CH₂CH₂CH₂CH=CHCH₂ + CH₂OH CH₂CH₂CH₂CH: Part 2: Br CHCH₂ Draw the missing resonance contributor. OCH3 + CH3CH₂CH₂CH=CHCH₂ CH3CH₂CH₂CH=CH-CH₂ + Br Br OCH 3 H₂CH₂CH₂CH=CH₂ view structure + + Br HBr Xarrow_forwardIdentify the dominant elimination mechanism (E1 or E2) for the following reaction. NaOH Neither Е2 Е1arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY