
(a)
Interpretation:
The configuration of the substitution products formed from the given reaction has to be drawn.
Concept Introduction:
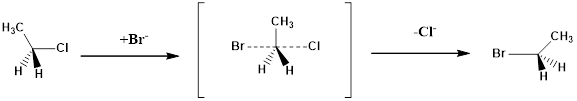
Structure of the substrate plays a major role in
Solvolysis: The reaction is defined as solvolysis reaction if solvent acts as nucleophile in the given reaction.
Configuration of a molecule: The configurations of a molecule arise due to the spatial arrangement of atoms. The configuration can be assigned by following CIP rules as follows.
Assign numbering to the groups which are bonded to the chiral carbon based on the molecular weight and electronegativity.
If the sequence of the numbering follows clockwise direction the chiral atom is assigned as R configuration.
If the sequence of the numbering follows anticlockwise direction the chiral atom is assigned as S configuration.
(b)
Interpretation:
The configuration of the substitution products formed from the given reaction has to be drawn.
Concept Introduction:
Primary carbocation < secondary carbocation < tertiary carbocation
Transition State: The state which defines the highest potential energy with respect to reaction co-ordinate between reactant and product. It is usually denoted by using the symbol ‘≠’.
Nucleophile: donates pair of electrons to positively charged substrate resulting in the formation of
Configuration of a molecule: The configurations of a molecule arise due to the spatial arrangement of atoms. The configuration can be assigned by following CIP rules as follows.
Assign numbering to the groups which are bonded to the chiral carbon based on the molecular weight and electronegativity.
If the sequence of the numbering follows clockwise direction the chiral atom is assigned as R configuration.
If the sequence of the numbering follows anticlockwise direction the chiral atom is assigned as S configuration.
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Essential Organic Chemistry, Global Edition
- Draw the major organic product of the SN1SN1 reaction:arrow_forwardRank the following compounds from greatest tendency to least tendency to undergo nucleophilic aromatic substitution: chlorobenzene 1-chloro-2,4-dinitrobenzene p-chloronitrobenzene a. Rank the same compounds from greatest tendency to least tendency to undergo electrophilic aromatic substitution.arrow_forwardDetermine and draw the structure of compound A. Then, determine the reactant/reagent needed to accomplish the one step transformation from benzene to A. Br₂ FeBr3 H (acid workup) PCC A ?arrow_forward
- Which benzene derivative would be the most reactive in an electrophilic aromatic substitution reaction? NO2arrow_forwardOffer an explanation for the observation that 4-chloropyridine is more reactive toward nucleophiles than 3-chloropyridine.arrow_forwardThe Reformatsky reaction is an addition reaction in which an organozinc reagent is used instead of a Grignard reagent to add to the carbonyl group ofan aldehyde or a ketone. Because the organozinc reagent is less reactive than a Grignard reagent, a nucleophilic addition to the ester group does not occur.The organozinc reagent is prepared by treating an a-bromo ester with zinc. Describe how each of the following compounds can be prepared, using a Reformatsky reaction:arrow_forward
- Hydroxy aldehydes A and B readily cyclize to form hemiacetals. Draw the stereoisomers formed in this reaction from both A and B. Explain why this process gives an optically inactive product mixture from A and an optically active product mixture from B.arrow_forwardWhat alkyl halide and nucleophile are needed to prepare each compound?arrow_forwardDraw the structure of the most stable enol tautomer(s)arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

