
CHEMISTRY-TEXT
8th Edition
ISBN: 9780134856230
Author: Robinson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 8.68SP
Bupropion, marketed as Wellbutr in, is a heavily prescribed medication used in the treatment of depression. Complete the following electron-dot structure for Bupropion by adding lone pairs of electrons.
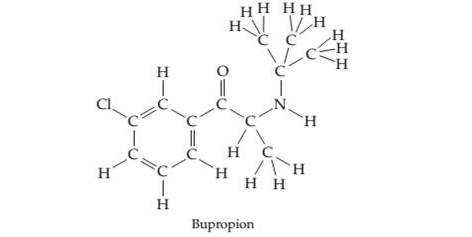
(a) How many
(b) Give the hybridization of each carbon atom in the molecule.
(c) Give the bond angles of each carbon atom.
(d) Give the hybridization of the N atom in the molecule.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The structure of caffeine is shown below.
(a) Complete the Lewis structure.
(b) How many pi bonds are present in caffeine? How many sigma bonds?
(c) Identify the hybridization of the carbon atoms.
(d) What is the value of the O-C-N angle?
In following decomposition reaction:2C3H8(g)⟶ C2H4(g)+ C3H6(g) + CH4(g) + H2(g)For each of the four carbon compounds, do the following:
(a) Draw Lewis structure.(b) Predict the geometry about the carbon atom.(c) Determine the hybridization of each type of carbon atom.
Azo dyes are organic dyes that are used for many applications,such as the coloring of fabrics. Many azo dyes arederivatives of the organic substance azobenzene, C12H10N2.A closely related substance is hydrazobenzene, C12H12N2.The Lewis structures of these two substances are given
(Recall the shorthand notation used for benzene.)(a) What is the hybridization at the N atom in each of thesubstances? (b) How many unhybridized atomic orbitalsare there on the N and the C atoms in each of the substances?(c) Predict the N—N—C angles in each of thesubstances. (d) Azobenzene is said to have greater delocalizationof its π electrons than hydrazobenzene. Discussthis statement in light of your answers to (a) and (b). (e) Allthe atoms of azobenzene lie in one plane, whereas thoseof hydrazobenzene do not. Is this observation consistentwith the statement in part (d)? (f) Azobenzene is an intensered-orange color, whereas hydrazobenzene is nearly colorless.Which molecule would be a better one to use in…
Chapter 8 Solutions
CHEMISTRY-TEXT
Ch. 8 - Prob. 8.1PCh. 8 - What is the number and geometric arrangement of...Ch. 8 - PRACTICE 8.3 Acetic acid, CH3CO2H , is the main...Ch. 8 - APPLY 8.4 Benzene, C6H6 , is a cyclic molecule in...Ch. 8 - PRACTICE 8.5 Identify the orbitals that overlap to...Ch. 8 - APPLY 8.6 Describe the bonding in propane, C3H8 ,...Ch. 8 - PRACTICE 8.7 Describe the hybridization of the...Ch. 8 - Describe the hybridization of each carbon atom in...Ch. 8 - Which orbitals overlap to form the sigma and pi...Ch. 8 - APPLY 8.10 Describe the hybridization of the...
Ch. 8 - Prob. 8.11PCh. 8 - Conceptual APPLY 8.12 Match the following...Ch. 8 - Prob. 8.13PCh. 8 - Prob. 8.14ACh. 8 - Prob. 8.15PCh. 8 - Prob. 8.16ACh. 8 - Prob. 8.17ACh. 8 - Prob. 8.18ACh. 8 - The B2 molecule has a MO diagram similar to that...Ch. 8 - Prob. 8.20ACh. 8 - PRACTICE 8.23 Draw two resonance structures for...Ch. 8 - APPLY 8.24 Draw two resonance structures for the...Ch. 8 - Prob. 8.23PCh. 8 - Prob. 8.24PCh. 8 - Caffeine is the most widely used stimulant...Ch. 8 - Prob. 8.26PCh. 8 - What is the geometry around the central atom in...Ch. 8 - What is the geometry around the central atom in...Ch. 8 - Three of the following molecular models have a...Ch. 8 - Identify each of the following sets of hybrid...Ch. 8 - The VSEPR model is a simple predictive tool that...Ch. 8 - The following ball-and-stick molecular model is a...Ch. 8 - The following ball-and-stick molecular model is a...Ch. 8 - Prob. 8.34CPCh. 8 - The dipole moment of methanol is =1.70D . Use...Ch. 8 - Methylarnine, CH3NH2 , is responsible for the odor...Ch. 8 - Prob. 8.37CPCh. 8 - Prob. 8.38SPCh. 8 - What shape do you expect for molecules that meet...Ch. 8 - How many charge clouds are there around the...Ch. 8 - Prob. 8.41SPCh. 8 - What shape do you expect for each of the following...Ch. 8 - What shape do you expect for each of the following...Ch. 8 - What shape do you expect for each of the following...Ch. 8 - Prob. 8.45SPCh. 8 - Prob. 8.46SPCh. 8 - What shape do you expect for each of the following...Ch. 8 - What bond angles do you expect for each of the...Ch. 8 - What bond angles do you expect for each of the...Ch. 8 - Acrylonitrile is used as the starting material for...Ch. 8 - Predict values for all bond angles in dimethyl...Ch. 8 - Oceanographers study the mixing of water masses by...Ch. 8 - A potential replacement for the chlorofluorocarbon...Ch. 8 - Explain why cyclohexane, a substance that contains...Ch. 8 - Like cyclohexane (Problem 8.54), benzene also...Ch. 8 - Use VSEPR theory to answer the following...Ch. 8 - Draw an electron-dot structure for each of the...Ch. 8 - What is the difference in spatial distribution...Ch. 8 - The average CC bond dissociation energy (D) is 350...Ch. 8 - What hybridization do you expect for atoms that...Ch. 8 - What spatial arrangement of charge clouds...Ch. 8 - What hybridization would you expect for the...Ch. 8 - What hybridization would you expect for the...Ch. 8 - Oxaloacetic acid is an intermediate involved in...Ch. 8 - The atoms in the amino acid glycine are connected...Ch. 8 - Describe the hybridization of the carbon atom in...Ch. 8 - Describe the hybridization of each carbon atom in...Ch. 8 - Bupropion, marketed as Wellbutr in, is a heavily...Ch. 8 - Efavirenz, marketed as Sustiva, is a medication...Ch. 8 - What is the hybridization of the B and N atoms in...Ch. 8 - Prob. 8.71SPCh. 8 - Aspirin has the following connections among atoms....Ch. 8 - The cation [HCNXeF]+ is entirely linear. Draw an...Ch. 8 - Acrylonitrile (C3H3N) is a molecule that is...Ch. 8 - The odor of cinnamon oil is due to cinnamaldehyde,...Ch. 8 - The following molecular model is a representation...Ch. 8 - Prob. 8.77SPCh. 8 - Which of the following substances would you expect...Ch. 8 - Which of the following substances would you expect...Ch. 8 - Why is the dipole moment of SO2 1.63 D hut that of...Ch. 8 - Prob. 8.81SPCh. 8 - The class of ions PtX42 , where X is a halogen,...Ch. 8 - Prob. 8.83SPCh. 8 - Prob. 8.84SPCh. 8 - Prob. 8.85SPCh. 8 - Prob. 8.86SPCh. 8 - Prob. 8.87SPCh. 8 - What are the most important kinds of...Ch. 8 - Of the substances Xe, CH3Cl , and HF which has:...Ch. 8 - Methanol (CH3OH;bp=65C) boils nearly 230 °C higher...Ch. 8 - Prob. 8.91SPCh. 8 - Prob. 8.92SPCh. 8 - Prob. 8.93SPCh. 8 - A liquid sample contains methylamine (CH3NH2)...Ch. 8 - Prob. 8.95SPCh. 8 - What is the difference in spatial distribution...Ch. 8 - Prob. 8.97SPCh. 8 - Use the MO energy diagram in Figure 8.22b to...Ch. 8 - Use the MO energy diagram in Figure 8.22 a to...Ch. 8 - The C2 molecule can be represented by an MO...Ch. 8 - Prob. 8.101SPCh. 8 - Prob. 8.102SPCh. 8 - Prob. 8.103SPCh. 8 - Draw a molecular orbital energy diagram for Li2 ....Ch. 8 - Calcium carbide, CaC2 , reacts with water to...Ch. 8 - At high temperatures, sulfur vapor is...Ch. 8 - Carbon monoxide is produced by incomplete...Ch. 8 - Make a sketch showing the location and geometry of...Ch. 8 - Make a sketch showing the location and geometry of...Ch. 8 - Prob. 8.110MPCh. 8 - Prob. 8.111MPCh. 8 - Prob. 8.112MPCh. 8 - Prob. 8.113MPCh. 8 - Just as individual bonds in a molecule are often...Ch. 8 - Cyclooctatetraenedian ion, C8H82 , is an organic...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In each of the following molecules, a central atom is surrounded by a total of three atoms or unshared electron pairs: SnCl2, BCl3, SO2. In which of these molecules would you expect the bond angle to be less than 120? Explain your reasoning.arrow_forwardButadiene, C4H6, is a planar molecule that has the followingcarbon–carbon bond lengths: (a) Predict the bond angles around each of the carbon atoms and sketch the molecule. (b) From left to right, what is the hybridization of each carbon atom in butadiene? (c) The middle C—C bond length in butadiene (1.48 Å) is a little shorter than the average C—C single bond length (1.54 Å). Does this imply that the middle C—C bond in butadiene is weaker or stronger than the average C—C single bond? (d) Based on your answer for part (c), discuss what additional aspects of bonding in butadiene might support the shorter middle C—C bond.arrow_forwardOctocrylene is an ingredient found in topical sunscreens. It is a water-resistant molecule that helps protect skin against harmful UVA and UVB radiation. Octocrylene Please answer the following questions: (a) What is the hybridisation of each nonhydrogen atom? (b) Are there two unique configurations possible about the C=C double bond? Please explain your answer. (c) Which of the two C-C bonds indicated by the arrows would you expect to be shorte.? Please explain your answer.arrow_forward
- 7. Nitrogen is the central atom in each of the species given. (a) Draw the Lewis electron-dot structure for each of the species. + NO₂ NO₂ NO₂ (b) List the species in order of increasing bond angle. Justify your answer. (c) For NO₂ and NO₂, give the hybridization of the nitrogen atom in it. (d) Identify the only one of the species that dimerizes and explain what causes it to do so.arrow_forwardEthyl acetate, C4H8O2, is a fragrant substance used both as asolvent and as an aroma enhancer. Its Lewis structure is (a) What is the hybridization at each of the carbon atomsof the molecule? (b) What is the total number of valenceelectrons in ethyl acetate? (c) How many of the valence electronsare used to make s bonds in the molecule? (d) Howmany valence electrons are used to make p bonds? (e) Howmany valence electrons remain in nonbonding pairs in themolecule?arrow_forwardConsider the reaction BF3 + NH3 -> F3B-NH3 (a) Describe the changes in hybridization of the B and N atoms as a result of this reaction. (b) Describe the shapes of all the reactant molecules with their bond angles. (c) Draw the overall shape of the product molecule and identify the bond angles around B and N atoms. (d) What is the name of the bond between B and N. (e)Describe the bonding orbitals that make the B and F, B and N & N and H bonds in the product molecule.arrow_forward
- Butadiene, C4H6, is a planar molecule that has the followingcarbon–carbon bond lengths:(a) Predict the bond angles around each of the carbon atomsand sketch the molecule. (b) From left to right, whatis the hybridization of each carbon atom in butadiene?(c) The middle C¬C bond length in butadiene (1.48 Å) isa little shorter than the average C¬C single bond length(1.54 Å). Does this imply that the middle C¬C bond in butadieneis weaker or stronger than the average C¬C singlebond? (d) Based on your answer for part (c), discuss what additional aspects of bonding in butadiene might supportthe shorter middle C¬C bond.arrow_forward(a) Draw Lewis structures for ethane (C2H6), ethylene(C2H4), and acetylene (C2H2). (b) What is the hybridizationof the carbon atoms in each molecule? (c) Predict whichmolecules, if any, are planar. (d) How many π and σ bondsare there in each molecule?arrow_forwardAzo dyes are organic dyes that are used for many applications,such as the coloring of fabrics. Many azo dyes arederivatives of the organic substance azobenzene, C12H10N2.A closely related substance is hydrazobenzene, C12H12N2.The Lewis structures of these two substances are(Recall the shorthand notation used for benzene.)(a) What is the hybridization at the N atom in each of thesubstances? (b) How many unhybridized atomic orbitalsare there on the N and the C atoms in each of the substances?(c) Predict the N¬N¬C angles in each of thesubstances. (d) Azobenzene is said to have greater delocalizationof its p electrons than hydrazobenzene. Discussthis statement in light of your answers to (a) and (b). (e) Allthe atoms of azobenzene lie in one plane, whereas thoseof hydrazobenzene do not. Is this observation consistentwith the statement in part (d)? (f) Azobenzene is an intensered-orange color, whereas hydrazobenzene is nearly colorless.Which molecule would be a better one to use in asolar…arrow_forward
- For each statement, indicate whether it is true or false. (a) The greater the orbital overlap in a bond, the weaker the bond. [b] The greater the orbital overlap in a bond, the shorter the bond. [c] To create a hybrid orbital, you could use the s orbital on one atom with a p orbital on another atom. [d] Nonbonding electron pairs cannot occupy a hybrid orbital.arrow_forwardThe lactic acid molecule, CH3CH(OH)COOH, gives sourmilk its unpleasant, sour taste. (a) Draw the Lewis structurefor the molecule, assuming that carbon always forms fourbonds in its stable compounds. (b) How many π and howmany σ bonds are in the molecule? (c) Which CO bond isshortest in the molecule? (d) What is the hybridization ofatomic orbitals around the carbon atom associated withthat short bond? (e) What are the approximate bond anglesaround each carbon atom in the molecule?arrow_forwardWhich molecule in each pair has the greater dipole mo-ment? Give the reason for your choice.(a) ClO₂ or SO₂(b) HBr or HCl(c) BeCl₂ or SCl₂(d) AsF₃ or AsF₅arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY