
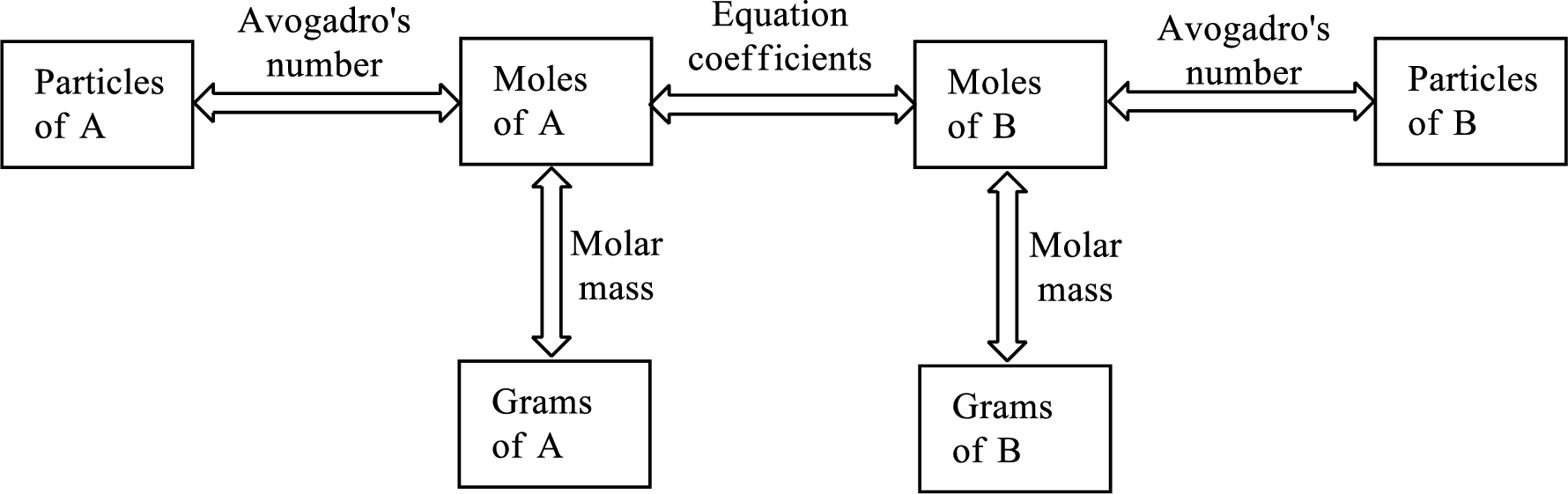
Interpretation:
Pathway for finding “moles of B” from “grams of A” using the conversion factors has to be chosen from the given options.
Concept Introduction:
Using the

Molar mass of a substance gives a relationship between the number of grams and number of moles of that particular substance.

The Chemical formula subscript also provides relationship between the number of moles of substance to the number of moles of its individual components present in the substance.

The moles of compound are related to moles of another compound by the coefficients present in the balanced chemical equation.
All the above relationship can be put into a single diagram as shown below,
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
General, Organic, and Biological Chemistry
- 3 attempts left Check my work Be sure to answer all parts. How many moles of aqueous magnesium ions and chloride ions are formed when 0.165 mol of magnesium chloride dissolves in water? moles magnesium ions moles chloride ions Prev 2 of 5arrow_forwardWhat is the percent yield when 32.18 g of octasulfur reacts with excess fluorine gas to produce 41.52 g hexaflouride. How much of the fluorine gas is consumed? S8 + F2 ---> SF6 Answer: Percent yield of SF6____________ Consumed F2________arrow_forwardAccording to the following reaction, how many moles of phosphoric acid will be formed upon the complete reaction of 0.282 moles diphosphorus pentoxide with excess water? diphosphorus pentoxide (s) + water (1) phosphoric acid (aq) moles phosphoric acid Submit Answer Try Another Version 1 item attempt remainingarrow_forward
- According to the following reaction, how many grams of nitrogen gas will be formed upon the complete reaction of 25.0 grams of hydrogen gas with excess nitrogen monoxide? nitrogen monoxide (g) + hydrogen (g) nitrogen (g) + water (I) grams nitrogen gas Submit Answer Try Another Version 3 item attempts remainingarrow_forward17.For the unbalanced equation: Mg3N2 + H2O -----------> Mg (OH) 2 + NH3 How many moles of Mg (OH) 2 are formed from 125g of H2O?Necessary answer. Unique option. A) 2.5 moles of Mg (OH) 2 B) 1.25 moles of Mg (OH) 2 C) 6.94 moles of Mg (OH) 2 D) 3.47 moles of Mg (OH) 2 E) None of the above.arrow_forwardIn the acetylene torch, acetylene gas (C2H2) burns in oxygen to produce carbon dioxide and water. 2C2H2 (g) + 5O2(g) à 4CO2(g) + 2H2O (g) Find the moles of CO2produced when 2.5 moles C2H2are burnt? Your Answer: Question 16 options: Answer unitsarrow_forward
- ___MgI2 +___ Br2 ➞ ___-MgBr2 +____ I2 Reaction Type:____________ When 560. g MgI2 reacts with an excess of Br2, how many moles of MgBr2will be formed? please show work in answerarrow_forwardMethane CHis a simple organic molecule that is used as a fuel—it is the major component of natural gas. The bottom of the ocean contains large amounts of methane hydrate (also called methane ice or methane clathrate), enough that it has been suggested as a major source of fuel. The methane hydrate contains 6 moles water for each mole of methane.What percentage of methane hydrate is methane? blank1 - Numeric Answer % What percentage is water? blank2 - Numeric Answer %arrow_forwardFor the reaction 2Na + 2H2O --> 2NaOH + H2 How many moles of H2 are produced from 3.36 moles of H20 ? Your Answer: Answerarrow_forward
- According to the following reaction, how many grams of nitrogen gas will be formed upon the complete reaction of 28.0 grams of ammonium nitrite? ammonium nitrite (aq) nitrogen (g) + water (1) grams nitrogen gas Submit Answer Try Another Version 2 Item attempts remainingarrow_forwardAccording to the following reaction, how many grams of calcium chloride will be formed upon the complete reaction of 28.7 grams of hydrochloric acid with excess calcium hydroxide? calcium hydroxide (aq) + hydrochloric acid (aq) >calcium chloride (aq) + water (1) grams calcium chloride Submit Answer Try Another Version 2 item attempts remainingarrow_forwardAccording to the following reaction, how many moles of carbon dioxide will be formed upon the complete reaction of 0.359 moles carbon monoxide with excess oxygen gas? carbon monoxide (g) + oxygen (g) >carbon dioxide (g) moles carbon dioxide Submit Answer Try Another Version 3 item attempts remainingarrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
