
Interpretation:
The pathway for solving a “grams of A” to “grams of B” by the use of conversion factor has to be chosen from the given options.
Concept Introduction:
Using the

Molar mass of a substance gives a relationship between the number of grams and number of moles of that particular substance.

The Chemical formula subscript also provides relationship between the number of moles of substance to the number of moles of its individual components present in the substance.

All the three above relationship can be put into a single diagram as shown below,
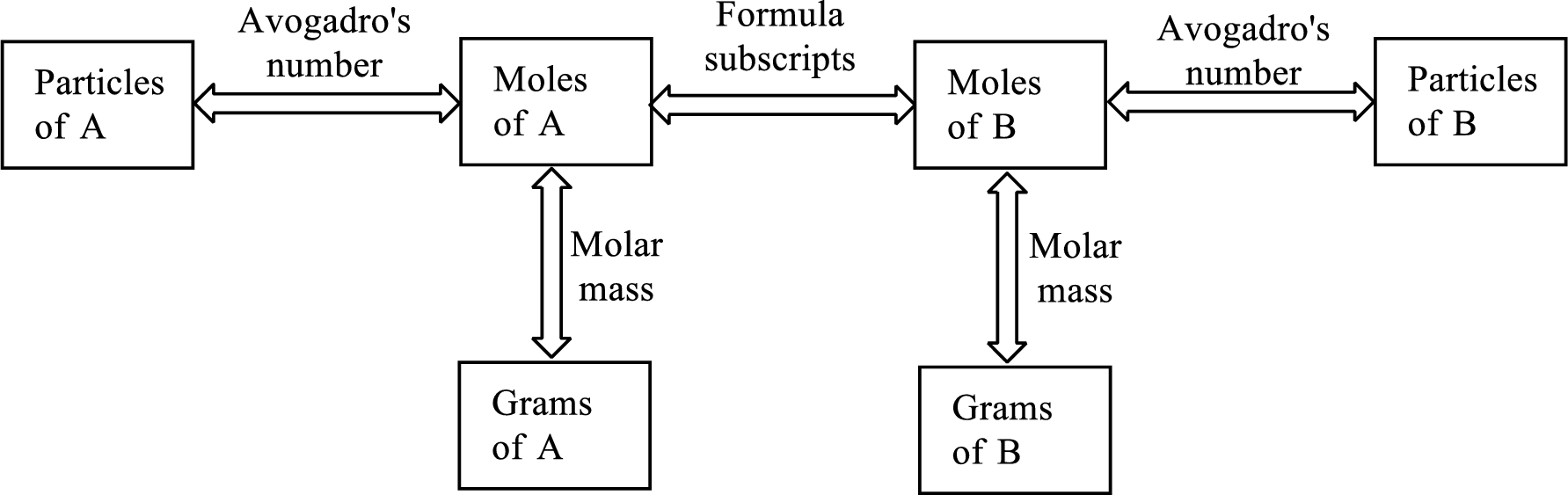
Trending nowThis is a popular solution!

Chapter 6 Solutions
General, Organic, and Biological Chemistry
- In the acetylene torch, acetylene gas (C2H2) burns in oxygen to produce carbon dioxide and water. 2C2H2 (g) + 5O2(g) à 4CO2(g) + 2H2O (g) Find the moles of CO2produced when 2.5 moles C2H2are burnt? Your Answer: Question 16 options: Answer unitsarrow_forwardHow many moles of N2 are needed to completely react with 5.65 mol of F2? N2 + 3F2---2NF3 Answer:arrow_forwardAccording to the following reaction, how many grams of nitrogen gas will be formed upon the complete reaction of 25.0 grams of hydrogen gas with excess nitrogen monoxide? nitrogen monoxide (g) + hydrogen (g) nitrogen (g) + water (I) grams nitrogen gas Submit Answer Try Another Version 3 item attempts remainingarrow_forward
- Q Search this cours References Use the References to access important values if needed for this question. According to the following reaction, how many grams of sodium iodide are required for the complete reaction of 29.6 grams of chlorine gas ? chlorine(g) + sodium iodide(s) sodium chloride(s) + iodine(s) grams sodium iodide Submit Answer chiometry of Rooctions: Gra. The is group attempt 1 of 5arrow_forwardProblems Mole.Mole and Mole.Mass.docx (22.7 KB) Directions: Answer each of the following questions using the equation provided. Be sure to balance each equation before solving any problems. Show all work for full credit. 1 point each for balancing each equation 1 point per question Problem Set 1: Cu + O, Cuo 1. If 101 grams of copper is used, how many moles of copper (II) oxide will be formed? 2. If 5.25 moles of copper are used, how many moles of oxygen must also be used? 3. If 78.2 grams of oxygen react with copper, how many moles of copper (II) oxide will be produced? Problem Set 2: С.Н. + O, CO, + H.O 4. How many moles of butane, C.H, are needed to react with 5.5 moles of oxygen? How many grams of carbon dioxide will be produced if 3.5 moles of O, react? 5. Prohlem Set 3: 54°F Sunny DELL F11 F12 F10 F9arrow_forwardWhat is the percent yield when 32.18 g of octasulfur reacts with excess fluorine gas to produce 41.52 g hexaflouride. How much of the fluorine gas is consumed? S8 + F2 ---> SF6 Answer: Percent yield of SF6____________ Consumed F2________arrow_forward
- According to the following reaction, how many grams of hydrofluoric acid will be formed upon the complete reaction of 26.1 grams of water with excess silicon tetrafluoride? silicon tetrafluoride (s) + water (1) hydrofluoric acid (aq) + silicon dioxide (s) grams hydrofluoric acid Submit Answer Try Another Version 1 item attempt remainingarrow_forwardmitted 30 The formula for iron (II) carbonate is FeCO3. 1. How many GRAMS of Fe2+ are present in 2.10 moles of iron (II) carbonate? 991 C 2. How many MOLES of CO32- ion are present in 3.20 grams of iron (II) carbonate? 0.00881 Submit Answer $ 4 888 F4 R FL % 5 V Retry Entire Group T Cengage Learning Cengage Technical Support G A 6 MacBook Air S B 15 F FG 9 more group attempts remaining Y ences] access important values if needed for this question. H & 7 4 F7 U N 8 J FB 1 M ( 9 K D FO O ) O I H L A F10 P command : grams. Email Instructor 8 ; moles. Previous { Save and Exit ? option 912 11 1 delete ? returnarrow_forwardAccording to the following reaction, how many grams of calcium chloride will be formed upon the complete reaction of 28.7 grams of hydrochloric acid with excess calcium hydroxide? calcium hydroxide (aq) + hydrochloric acid (aq) >calcium chloride (aq) + water (1) grams calcium chloride Submit Answer Try Another Version 2 item attempts remainingarrow_forward
- According to the following reaction, how many moles of phosphoric acid will be formed upon the complete reaction of 0.282 moles diphosphorus pentoxide with excess water? diphosphorus pentoxide (s) + water (1) phosphoric acid (aq) moles phosphoric acid Submit Answer Try Another Version 1 item attempt remainingarrow_forwardGaseous ethane (CH,CH,) reacts with gaseous oxygen gas (02) to produce gaseous carbon dioxide (CO,) and gaseous water (H,0). If 31.4 g of water is produced from the reaction of 29.8 g of ethane and 73.2 g of oxygen gas, calculate the percent yield of water. 圖 Round your answer to 3 significant figures. do % Ar Explanation Checkarrow_forward2:41 1 LTE 1 Search Question 18 of 24 Submit According to the balanced reaction below, calculate the moles of NH3 that form when 4.2 mol of N2H4 completely reacts N2H4(1) → 4 NH3(g) + N2(g) STARTING AMOUNT ADD FACTOR ANSWER RESET *( ) 32.06 4.2 mol NH3 3.2 5.6 g N2H4 3 16.8 mol N2 6.022 x 1023 g N2 28.02 mol N2H4 17.04 g NH3 4 1 Tap here or pull up for additional resourcesarrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
