
(a)
Interpretation:
Using the recommended daily dietary intake of calcium the amount of calcium required for adult men and premenopausal women should be identified.
Concept Introduction:
Importance of Calcium in diet:
- 1. Calcium is required by human body for muscle contraction, hormones release and transmission of messages via nerves.
- 2. Correct intake of calcium helps one from osteoporosis which occurs as density of the bone decreases and results in structural defect and weakness of bones.
(b)
Interpretation:
The mass of calcium citrate required to attain the recommended daily intake of calcium should be calculated.
Concept Introduction:
Calcium citrate: It is recommended for calcium deficiency diseases namely rickets, tetany and osteoporosis. The molecular formula of Calcium citrate is
Importance of Calcium in diet:
- 1. Calcium is required by human body for muscle contraction, hormones release and transmission of messages via nerves.
- 2. Correct intake of calcium helps one from osteoporosis which occurs as density of the bone decreases and results in structural defect and weakness of bones.
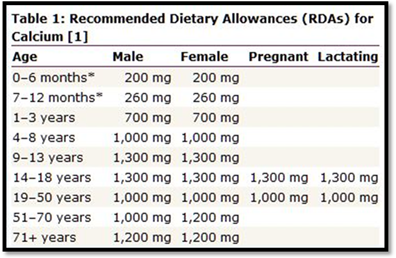
The table that depicts recommended daily intake of calcium is as follows,
Moles: One mole is equivalent to the mass of the substance consists same number of units equal to the atoms present in
The mole is actually quantity of particles that is
The sum of mass of all atoms present in formula of chemical substance is referred as molecular or formula weight of that substance.
Mass: It is the quantitative measure of a substance. The amount of matter present in substance is expressed as mass. The
Molar mass: It is obtained by dividing the mass of substance with the amount of substance and the S.I. unit of molar mass is
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- Biochemistry Please help. Thank you When carbamyl phosphate is joined to L-ornathine, where does the energy for the reaction come from?arrow_forwardBiochemistry Question Please help. Thank you What is the function of glutamate dehydrogenase?arrow_forwardBiochemistry Question Please help. Thank you How and why does a high protein diet affect the enzymes of the urea cycle?arrow_forward
- Biochemistry What is the importance of the glucose-alanine cycle?arrow_forwardBiochemistry Assuming 2.5 molecules of ATP per oxidation of NADH/(H+) and 1.5molecules of ATP per oxidation of FADH2, how many ATP are produced per molecule of pyruvate? Please help. Thank youarrow_forward1. How would you explain the term ‘good food’? 2. How would you define Nutrition? 3. Nutrients are generally categorised into two forms. Discuss.arrow_forward
- Biochemistry Question. Please help solve. Thank you! Based upon knowledge of oxidation of bioorganic compounds and howmuch energy is released during their oxidation, rank the following, from most to least, with respect to how much energy would be produced from each during their oxidation. Explain your placement for each one.arrow_forwardBiochemistry Question.For the metabolism of amino acids what is the first step for theirbreakdown? Why is it necessary for this breakdown product to be transported to the liver? For the catabolism of the carbon backbone of these amino acids, there are 7 entry points into the “standard” metabolic pathways. List these 7 entry points and which amino acids are metabolized to these entry points. Please help. Thank you!arrow_forwardBiochemistry Question. Please help. Thank you. You are studying pyruvate utilization in mammals for ATP production under aerobic conditions and have synthesized pyruvate with Carbon #1 labelled with radioactive C14. After only one complete cycle of the TCA cycle, which of the TCA cycle intermediates would be labeled with C14? Explain your answer. Interestingly, you find C14 being excreted in the urine. How does it get there?arrow_forward
- Biochemistry question. Please help with. Thanks in advance For each of the enzymes listed below, explain what the enzyme does including function, names (or structures) of the substrate and products and the pathway(s) (if applicable) it is/are found in. (a) ATP synthetase (b) succinate dehydrogenase (c) isocitrate lyase (d) acetyl CoA carboxylase (e) isocitrate dehydrogenase (f) malate dehydrogenasearrow_forwardDraw and name each alcohol and classify it as primary, secondary, or tertiary. Explain your answer thoroughly.arrow_forwardDraw the product of each reaction. If there are multiple products, draw only the major product. Explain your answer thoroughly.arrow_forward
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning





