
Concept explainers
To analyze:
The ratio of Saturated acid to unsaturated fatty acid in the ethanolamine phospholipids of an animal, given that the fluorescence of membrane probe is 0.27.
Given:
Fluorescence of membrane probe is 0.27. The fluorescence of the membrane probes from different animals was recorded in Figure 1 as a graph. The graph represents the fluorescence with respect to a set of temperature ranging from 0°C to 37°C.
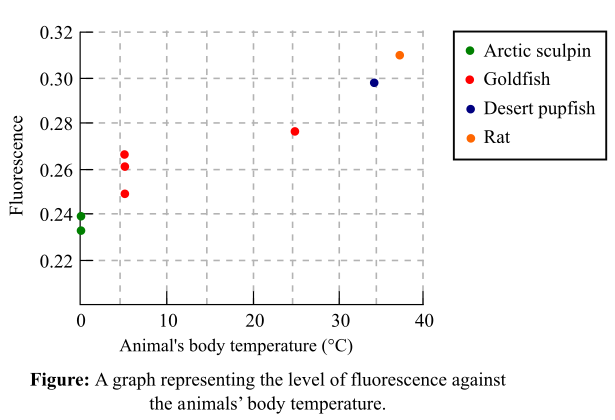
Figure 1: A graph representing the level of fluorescence against the animal’s body temperature.
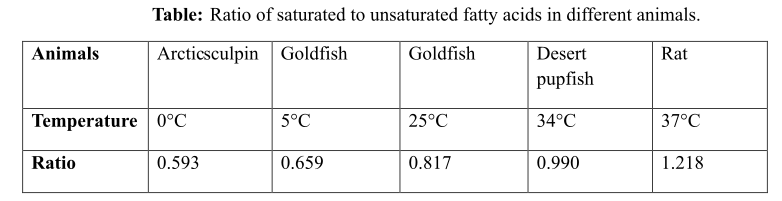
The ratio of saturated to unsaturated fatty acids in different animals, against a particular temperature is provided in the following Table 1 below:
Table 1: The ratio of saturated to unsaturated fatty acids in different animals
Introduction:
The saturated fatty acids are those that have single bonds in hydrocarbon chains, unlike the unsaturated fatty acids that have double or triple bonds in their hydrocarbon chains. The ethanolamine is a phospholipid that is generally present in the inner leaflet of the plasma membrane. The hydrocarbon chain of the ethanolamine phospholipid contains more concentration of unsaturated fatty acids when the temperature of the membrane is low. The ratio of saturated to unsaturated fatty acids is used by the researchers to find out the flexibility and the composition of the plasma membrane. The information also provides the range of the temperatures at which the organisms can operate their biological functions.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Life: The Science of Biology
- a. You selectively label phospholipids with a fluorescent dye and perform the FRAP assay. You detect a single fluorescence molecule move 100 micrometre into the photobleached area in 2.5 seconds. What is the diffusivity constant for this phospholipid? b. Increasing the ratio of unsaturated to saturated phospholipids in a biological membrane increases membrane fluidity. Describe how the FRAP photobleaching and recovery curves would differ between membranes with 63% and 41% unsaturated phospholipids. c. Draw the FRAP photobleaching and recovery curves for your answer in b. d. What does the “immobile fraction” in a FRAP experiment suggest (see diagram)?arrow_forwardSuppose the major solutes in intact lysosomes are KCl (~0.1 M) and NaCl (~0.03 M). When isolating lysosomes, what concentration of sucrose is required in the extracting solution at room temperature (25oC) to prevent swelling and lysis? Calculate how much sucrose (342.3 g/mol) would you need to make 500 ml of this extracting solution.arrow_forwardSuppose the major solutes in intact lysosomes are KCl (~0.1 M) and NaCl(~0.03 M). When isolating lysosomes, what concentration of sucrose isrequired in the extracting solution at room temperature (25 °C) to preventswelling and lysis?arrow_forward
- Calculate the energy cost (free-energy change) of pumping Ca2+ from thecytosol, where its concentration is about 1.0 × 10−7 M, to the extracellularfluid, where its concentration is about 1.0 mM. Assume a temperature of 37°C (body temperature in a mammal) and a standard transmembrane potentialof 50 mV (inside negative) for the plasma membrane.arrow_forwardA researcher is isolating proteins from a cell via different techniques. They find that when the cell is ground with a blender, all of the proteins in the cell can be extracted. However if instead the cell membranes are agitated in a 1.2 M KCl mixture, only about 70% of the total protein is extracted.What class of membrane proteins are in this soluble extract and what forces normally hold them to the membrane?What class of membrane proteins are the 30% of proteins that remain behind, and what forces hold these proteins in the membrane? If a solution of ionic solids was not available, what other solvent systems could be used to extract these proteins?arrow_forwardyeasts are able to produce high internal concentrations of glycerol to counteract the osmotic pressure of the surrounding media. suppose that a sample of yeast cells were placed in a 4% sodium chloride solution by weight. The density of solution is at 25 C = 1.02 g/ml, Molecular weight of solute = 58.44 g/mol, i of glycerol = 1 and R=0.08205 L-atm/mol-K What is the weight of solute in grams What is the moles of solute What is the volume of the solution in liters What is the molarity of the solution What is the value of the temperature to be used to solved for the osmotic pressure of the solution What is the osmotic pressure of solutionarrow_forward
- How many copies of a protein need to be present in a cell in order for it to be visible as a band on an SDS gel? Assume that you can load 100 µg of cell extract onto a gel and that you can detect 10 ng in a single band by sil ver staining the gel. The concentration of protein in cells is about 200 mg/mL, and a typical mammalian cell has a volume of about 1000 μm³ and a typical bacterium a vol ume of about 1 µm³. Given these parameters, calculate the number of copies of a 120-kd protein that would need to be present in a mammalian cell and in a bacterium in order to give a detectable band on a gel. You might try an order-of-magnitude guess before you make the calcula tions.arrow_forwardA spherical cell with the diameter of 10uMhas a protein concentration of 20 mg/ml. Determine the number of protein molecules within the cell if the molecular weight of an average protein is 50,000 daltons (g/mol). Recall that Avogadro's number is N₁ = 6.0221367×1023 molecules/mol.arrow_forwardMembranes that contain a significant proportion of cis unsaturated fatty acids are more fluid than similar membraneswith higher levels of saturated fatty acids. Explain.arrow_forward
- DO NOT COPY THE ANSWER FROM THE SAME QUESTION. Sassa, a biology professor, wanted to demonstrate to her students the applicability of a dialyzing membrane (DM) as a model for the cell membrane by enclosing an aqueous solution in a DM bag and immersing in a beaker containing a different solution. Substances available which are permeable to the DM include 0.02 M NaCl, 0.03 M glucose, and 0.01 M glucose. The only substance available which is completely impermeable to the DM is the 0.01 M lactose. Using the substances given and materials such as a beaker, stirring rod, and string, draw ONLY ONE set-up that can be demonstrated by Ms. Sassa that will satisfy ALL of the following conditions:a. No solute will exhibit a net diffusion out of the cell.b. Glucose will exhibit a net diffusion into the cell.c. NaCl will exhibit a zero net d. No net movement of lactose from the inside to outside of the cell. Make sure to label properly the substance inside the beaker and inside the DM bag.arrow_forwardin your own words, describe what would haooeb to the phospholipids f the plasma membrane in animals cells if cholesterol was not rpesent and the cell was expoed to extremely low temperatures?arrow_forwardMembrane lipid molecules exchange places with their lipid neighbors every 10–7 second. A lipid molecule diffuses from one end of a 2-μm-long bacterial cell to the other in about 1 second. Are these two numbers in agreement (assume that the diameter of a lipid head group is about 0.5 nm)? If not, can you think of a reason for the difference?arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





