
CAMPBEL BIOLOGY:CONCEPTS & CONNECTIONS
10th Edition
ISBN: 9780136538820
Author: Taylor
Publisher: INTER PEAR
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6, Problem 15TYK
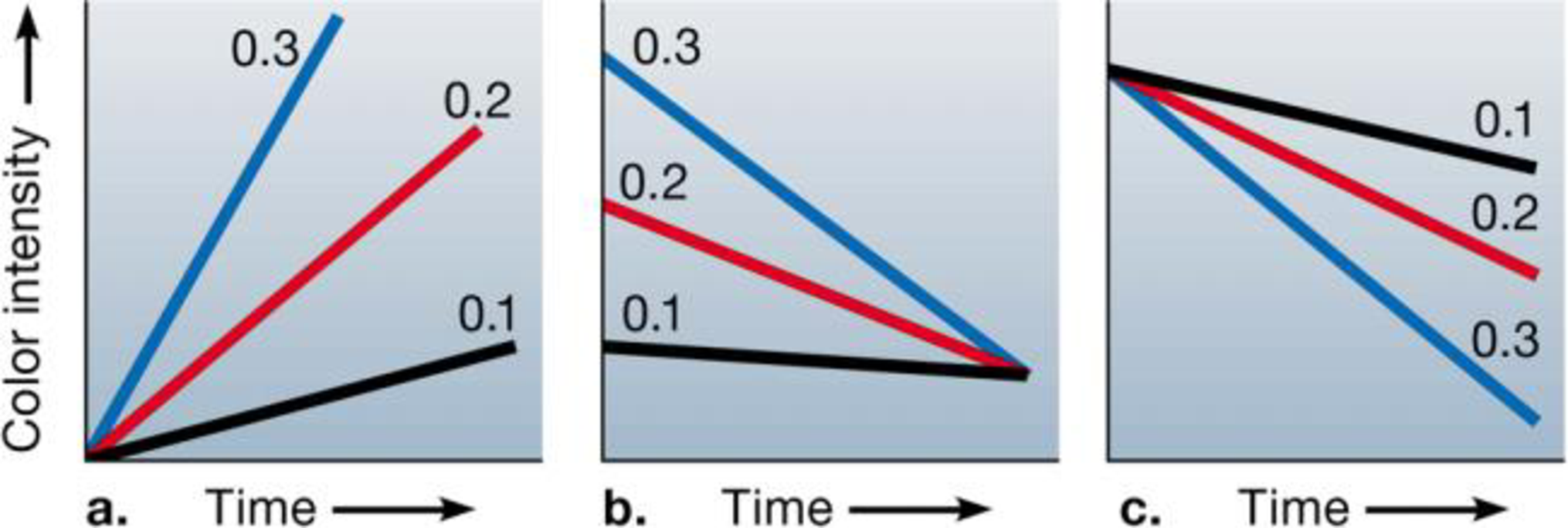
In the citric acid cycle, an enzyme oxidizes malate to oxaloacetate, with the production of NADH and the release of H+. You are studying this reaction using a suspension of bean cell mitochondria and a blue dye that loses its color as it takes up H+. You set up reaction mixtures with mitochondria, dye, and three different concentrations of malate (0.1 mg/L, 0.2 mg/L, and 0.3 mg/L). Which of the following graphs represents the results you would expect, and why?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The enzyme malate dehydrogenase catalyzes the conversion of malate to oxaloacetate in the final step that completes the Citric Acid Cycle. The ∆G of this reaction is +29.7 kJ/mol. Explain how this thermodynamically unfavorable reaction could possibly be accomplished by the cell even though the reaction is not coupled to a favorable reaction.
Enzymes
Calculate the AG between glucose and ATP catalyzed by hexokinase:
glucose + ATP glucose-6-phosphate + ADP
Is the reaction spontaneous?
Given data:
Glucose-6-phosphate + H,O glucose + P,
AG' = -3138 cal/mole
ATP+H,O=ADP + P:
AG'= -7700 cal/mole
In the energy recovery phase of glycolysis, Glyceraldehyde 3-p reacts with NAD+
In this reaction, which will be oxidized, and which will be reduced. In simple terms, what does it mean when a chemical is oxidized, and what does it mean when a chemical is reduced?
Chapter 6 Solutions
CAMPBEL BIOLOGY:CONCEPTS & CONNECTIONS
Ch. 6 - Fill in the blanks in this summary map to help you...Ch. 6 - A biochemist wanted to study how various...Ch. 6 - In glycolysis, _____ is oxidized and _____ is...Ch. 6 - Most of the CO2 from cellular respiration is...Ch. 6 - Which of the following is the most immediate...Ch. 6 - Which of the following is a true distinction...Ch. 6 - The poison cyanide binds to an electron carrier...Ch. 6 - In which of the following is the first molecule...Ch. 6 - Which of the three stages of cellular respiration...Ch. 6 - Compare and contrast fermentation as it occurs in...
Ch. 6 - Prob. 11TYKCh. 6 - Prob. 12TYKCh. 6 - Prob. 13TYKCh. 6 - Oxidative phosphorylation involves the flow of...Ch. 6 - In the citric acid cycle, an enzyme oxidizes...Ch. 6 - ATP synthase enzymes are found in the prokaryotic...Ch. 6 - Prob. 17TYKCh. 6 - For a short time in the 1930s, some physicians...Ch. 6 - Explain how the mechanism of brown fat metabolism...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- The enzyme aldolase catalyzes the reaction shown in the glycolytic pathway: Fructose 1,6-bisphosphate dihydroxyacetone phosphate + glyceraldehyde 3-phosphate The AG" for the reaction is +23.8 kJ mol¯¹ (+5.7 kcal mol−¹), whereas the AG in the cell is −1.3 kJ mol¯¹ (−0.3 kcal mol¯¹). Calculate the ratio of products to reactants under standard (equilibrium) conditions at 37°C. [products] [reactants] 7 x10-5 [products] [reactants] Incorrect Aldolase ===== Calculate the ratio of products to reactants under intracellular conditions at 37°C. 4 ×10-5 Incorrect Complete the statement using your results. under standard conditions under intracellular conditions A reaction that is endergonic under standard conditions can be converted into an exergonic reaction by maintaining the ratio of products to reactants below the equilibrium value.arrow_forwardInterested in studying ethanol fermentation, you prepare two solutions of yeast extract that contains the cytosolic contents of Saccharomyces cerevisiae. You add glucose to both extracts at time zero, but to one (in red) you also add inorganic phosphate (added in two batches). Using a gas sensor, you monitor the CO2 generated by the two solutions over time and obtain the following data:arrow_forwardThe AG of the reaction C6H12O6 + 60₂ --> 6CO₂ + 6H₂O is -686 kcal/mol glucose oxidized. The AG of the reaction ADP + P₁ --> ATP + H₂O is + 7.3 kcal/mol ATP synthesized. The oxidation of glucose can be coupled to the synthesis of ATP. If the coupling is 50% efficient, how many molecules of ATP can be synthesized per molecule of glucose oxidized? Round your answer to the nearest whole number.arrow_forward
- Decylic acid is a saturated fatty acid that occurs naturally in coconut oil and palm kernel oil. Calculate the net ATP yield when decylic acid undergoes complete B oxidation. The formula of decylic acid is shown below: (Given: The oxidation of one NADH yields 2.5 ATP; the oxidation of one FADH2 yields 1.5 ATP; and the oxidation of one acetyl CoA yields 10 ATP.) O 50 ATP O 52 ATP 66 ATP OH O 64 ATParrow_forwardPhotosynthesis is defined as the chemical process, wherein carbon dioxide in the presence of water and radiant energy gets converted to glucose (chemical energy), giving out oxygen as byproducts. In the equation, the combining reactants and resulting products are expressed along with their respective numbers of molecules. Putting this in a simplified formula, the following equation represents this process. CO2 + H2O + Light energy → C6H12O6 + O2 Considering this equation, how plant lovers (plantitos and plantitas) able to cultivate their plants?arrow_forwardWhat is the reduction potential (DE) for the reaction catalyzed by isocitrate dehydrogenase if the citric acid cycle is fluxing? To get the answer scored correctly: Please report the answer to three decimal places. Example: 0.123 or -0.111 Do not include units.arrow_forward
- Starting with glucose, there are five steps in the EMP/TCA metabolic pathways that produce NADH (which is converted to ATP or energy). Use class notes, Shuler, Kargi and DeLisa book, or the internet. 1. A. Draw the chemical structures of the reactant(s) and product(s); as well as a balanced chemical reaction for the step in the TCA (Krebs) cycle where isocitrate is converted to a-ketoglutarate (step 3 in the class notes). (You don't need to draw the structures of coenzyme A (COA), NAD* or NADH.) B. What enzymes catalyze steps 3 and 5 in the Kreb's cycle from the class notes? C. Would you categorize step 8 in the TCA cycle where malate is converted to oxaloacetate as an oxidation or reduction of malate? D. Considering that some energy is spent transporting acetyl-CoA to mitochondria, how much net energy (in kcal) is produced by converting 15 mol of glucose to CO₂ in the glycolysis & Krebs cycle pathways? E. How many kcals of energy from oxidizing 15 mol of glucose to CO₂ (your answer to…arrow_forwardThe complex chemical reactions that occur in living organisms can sometimes be summarized with chemical equations. The overall process of photosynthesis and aerobic respiration can both be summarized in this way. For the following equations, first determine whether each is summarizing photosynthesis or aerobic respiration, and then click on each of the boxes below the equations and correctly label all of the reactants and products. The following equation summarizes the reactants and products of A. aerobic respiration B. Photosynthesisarrow_forwardInterested in studying ethanol fermentation, you prepare two solutions of yeast extract that contains the cytosolic contents of Saccharomyces cerevisiae. You add glucose to both extracts at time zero, but to one (in red) you also add inorganic phosphate (added in two batches). Using a gas sensor, you monitor the CO2 generated by the two solutions over time and obtain the following data: a. Why is more CO2 generated upon the addition of phosphate? b. You become worried that the flasks would burst open to the build up in gas pressure. What other molecules could you add to the extract to slow down CO2 generation even in the presence of glucose and Pi?arrow_forward
- The complete combustion of palmitate and glucose yields 9781 kJ ∙ mol−1 and 2850 kJ ∙ mol−1 of free energy, respectively. Compare these values to the free energy (as ATP) obtained though catabolism of palmitate and glucose under standard conditions. Which process is more efficient?arrow_forwardWhat terms would best describe the above coupled reaction? (If the DGo for ATP hydrolysis into ADP + inorganic phosphate is -7.3 kcal/mole, and the DGo for maltose synthesis from glucose + glucose is +3.7 kcal/mole, calculate the standard free energy change for the combined reaction of ATP + glucose + glucose g ADP + maltose + inorganic phosphate.) it is non-spontaneous and endothermic (because the overall DGo is negative) it is spontaneous and exothermic (because the overall DGo is negative) it is non-spontaneous and endothermic (because the overall DGo is positive) it is spontaneous and exothermic (because the overall DGo is positive) it is non-spontaneous and exothermic (because the overall DGo is negative)arrow_forwardThe formation of ATP by ATP Synthase is part of a coupled reaction, as discussed in class. Given the information below and using what you know about ATP Synthase and the proton motive force, calculate the overall AG and determine if the coupled reaction is favorable overall. Note that you do not need to worry about intermediates like we did for the class example. ADP + Pi → ATP has a AG = 7.3 kcal/mol 2 moles of protons traveling back down their gradient across the inner mitochondrial membrane has a AG = -10.28 kcal/mol %3D O a. Overall AG = -17.58 kcal/mol, favorable overall O b. Overall AG = -2.98 kcal/mol, favorable overall %3D O c. Overall AG = 2.98 kcal/mol, unfavorable overall O d. Overall AG = 17.58 kcal/mol, favorable overallarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
Intro to Food Microbiology; Author: A professor pressing record;https://www.youtube.com/watch?v=vg8fSmk0dVU;License: Standard youtube license