
Concept explainers
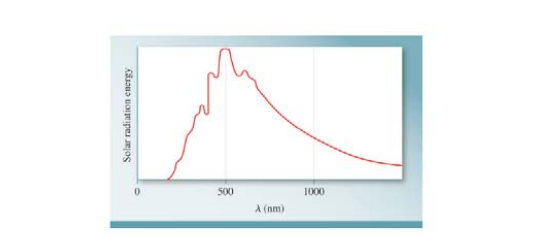
Blackbody radiation is the term used to describe the dependence of the radiation energy emitted by an object on wavelength at a certain temperature. Planck proposed the quantum theory to account for the dependence Shown in the figure is a plot of the radiation energy emitted by our sun versus wavelength This curve is characteristic of objects at about 6000 K. which is the temperature at the surface of the sun. At a higher temperature, the curve has a similar shape but the maximum will shift to a shorter wavelength (a) What does this curve reveal about two consequences of great biological significance on Faith? (b) How are astronomers able to determine the temperature at the surface of stars in general?
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Chemistry
Additional Science Textbook Solutions
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
Organic Chemistry
Elementary Principles of Chemical Processes, Binder Ready Version
Chemistry: Atoms First
Organic Chemistry As a Second Language: Second Semester Topics
- A baseball weighs 142 g. A professional pitcher throws a fast ball at a speed of 100 mph and a curve ball at 80 mph. What wavelengths are associated with the motions of the baseball? If the uncertainty in the position of the ball is 12 wavelength, which ball (fast ball or curve) has a more precisely known position? Can the uncertainty in the position of a curve ball be used to explain why batters frequently miss it?arrow_forward6.32 What are the mathematical origins of quantum numbers?arrow_forwardIn 1885, Johann Balmer, a mathematician, derived the following relation for the wavelength of lines in the visible spectrum of hydrogen =364.5 n2( n2 4) where in nanometers and n is an integer that can be 3, 4, 5, . . . Show that this relation follows from the Bohr equation and the equation using the Rydberg constant. Note that in the Balmer series, the electron is returning to the n=2 level.arrow_forward
- 6.99 The photoelectric effect can he used in engineering designs for practical applications. For example, infrared goggles used in night-vision applications have materials that give an electrical signal with exposure to the relatively long wavelength IR light. If the energy needed for signal generation is 3.51020J , what is the wavelength and frequency of light that barely can be detected?arrow_forwardWhat is the wavelength of a proton traveling at a speed of 6.21 km/s? What would be the region of the spectrum for electromagnetic radiation of this wavelength?arrow_forward6.93 A mercury atom is initially in its lowest possible (or ground state) energy level. The atom absorbs a photon with a wavelength of 185 nm and then emits a photon with a frequency of 4.9241014HZ . At the end of this series of transitions, the atom will still be in an energy level above the ground state. Draw an energy-level diagram for this process and find the energy of this resulting excited state, assuming that we assign a value of E = 0 to the ground state. (This choice of E = 0 is not the usual convention, but it will simplify the calculations you need to do here.)arrow_forward
- Investigating Energy Levels Consider the hypothetical atom X that has one electron like the H atom but has different energy levels. The energies of an electron in an X atom are described by the equation E=RHn3 where RH is the same as for hydrogen (2.179 1018 J). Answer the following questions, without calculating energy values. a How would the ground-state energy levels of X and H compare? b Would the energy of an electron in the n = 2 level of H be higher or lower than that of an electron in the n = 2 level of X? Explain your answer. c How do the spacings of the energy levels of X and H compare? d Which would involve the emission of a higher frequency of light, the transition of an electron in an H atom from the n = 5 to the n = 3 level or a similar transition in an X atom? e Which atom, X or H, would require more energy to completely remove its electron? f A photon corresponding to a particular frequency of blue light produces a transition from the n = 2 to the n = 5 level of a hydrogen atom. Could this photon produce the same transition (n = 12 to n = 5) in an atom of X? Explain.arrow_forwardAn FM radio station found at 103.1 on the FM dial broadcasts at a frequency of 1.031188s1 (103.1 MHz). What is the wavelength of these radio waves in meters?arrow_forwardPlanck originated the idea that energies can be quantized. What does the term quantized mean? What was Planck trying to explain when he was led to the concept of quantization of energy? Give the formula he arrived at and explain each of the terms in the formula.arrow_forward
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





