
Concept explainers
(a)
Interpretation:
The point on the graph corresponds to the reactants has to be given.
The given graph is,
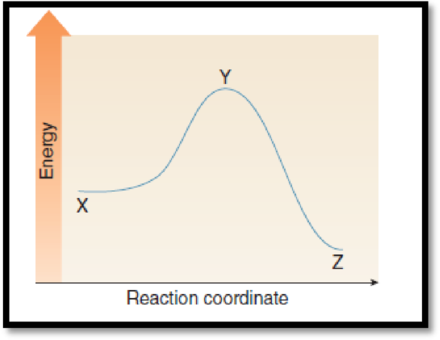
Figure 1
(b)
Interpretation:
The point on the graph corresponds to the product has to be given.
The given graph is,
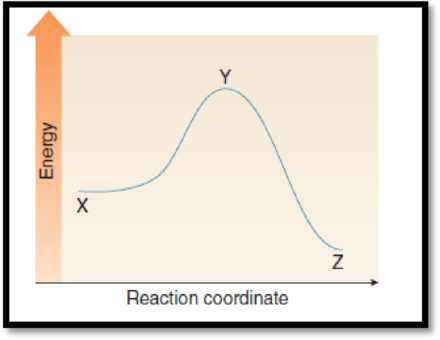
Figure 1
(c)
Interpretation:
The point on the graph corresponds to the transition state has to be given.
The given graph is,
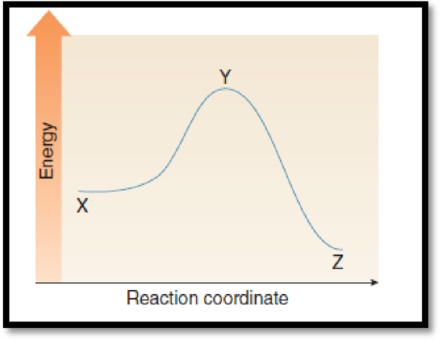
Figure 1
(d)
Interpretation:
The difference in energy between the two points which equals activation energy has to be given.
The given graph is,
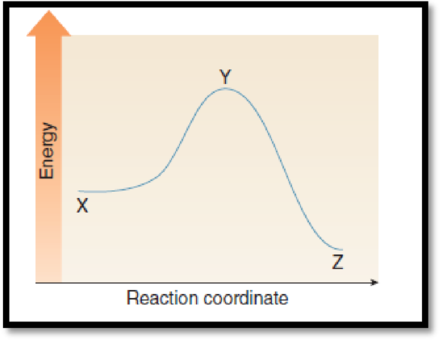
Figure 1
(e)
Interpretation:
The difference in energy between the two points which equals enthalpy has to be given.
The given graph is,
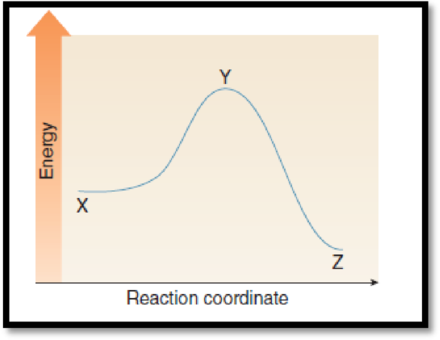
Figure 1
(f)
Interpretation:
The point with highest energy has to be given.
The given graph is,
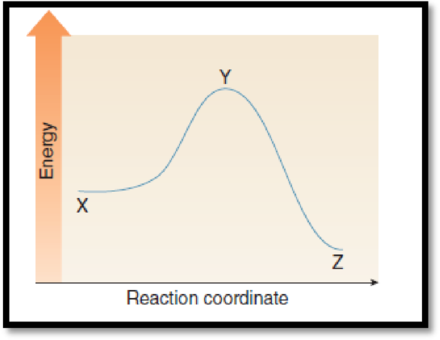
Figure 1
(g)
Interpretation:
The point with lowest energy has to be given.
The given graph is,
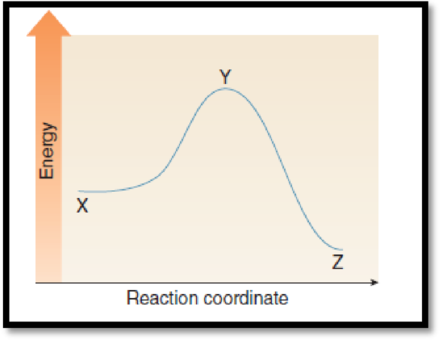
Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Principles of General, Organic, Biological Chemistry
- xplain why aluminum cans make good storage containers for soft drinks. Styrofoam cups can be used to keep coffee hot and cola cold. How can this be?arrow_forward9.75 Explain why each of the following chemical equations is not a correct formation reaction. (a) 4Al(s)+3O2(g)2Al2O3(s) (b) N2(g)+32H2(g)NH3(g) (c) 2Na(s)+O(g)Na2O(s)arrow_forward. A(n) _______ speeds up a reaction without being consumed.arrow_forward
- Explain why each of the following chemical equations is not a correct formation reaction: 4Al( s )+3 O 2 ( g )2 Al 2 O 3 ( s ) N 2 ( g )+ 3 2 H 2 ( g ) NH 3 ( g ) 2Na( s )+O( g ) Na 2 O( s )arrow_forwardYou have a large balloon containing 1.0 mol of gaseous water vapor at 80 C. How will each step affect the internal energy of the system? (a) The temperature of the system is raised to 90 C. (b) The vapor is condensed to a liquid, at 40 C.arrow_forward. Consider the exothermic reaction CO(g)+2H2(g)CH3OH(l)Predict three changes that could be made to the system that would decrease the yield of product over that produced by a system in which no change was made.arrow_forward
 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning





