
Concept explainers
To analyze:
The variations in Po2 (in units of mmHg) in a typical open-close-fluttering cycle. Also explain the estimated mm Hg Po2 in the atmosphere and inside the tubes of trachea in all the three given experiments shown in figure B. Here, 1 kPa (kilopascal) = 7.5 mm Hg.
Given information:
The microscopic plastic tubes were inserted into the tracheae of the pupae of Attacus atlas moths by the researchers to study the tracheal function in the moths. The microtubes were attached to the sensors for recording the rate of CO2 that was released from the tracheae and also the intratracheal concentrations of O2.
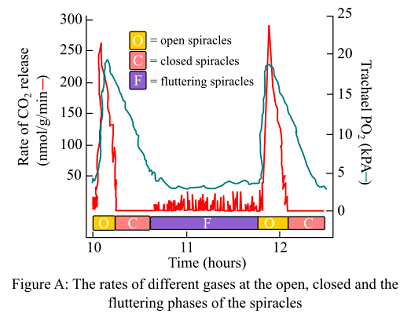
The pupae were placed in the chambers by the researchers and the normal atmospheric levels of the gases were maintained. Researchers then recorded the behavior of spiracles, rate of CO2 release, and O2 concentrations in the tracheae. The results are shown in figure A.
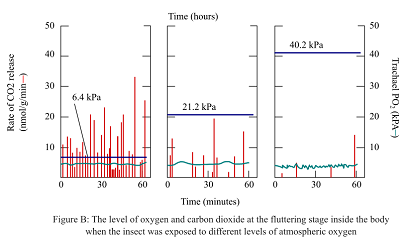
In order to examine the spiracles performance in the different atmospheric conditions and their effects on the rate of CO2 release and O2 concentrations, pupae were exposed to the different levels of atmospheric O2. The levels of the release of CO2 and O2 concentrations were calculated during the fluttering stage as shown in the figure B.
Introduction:
The insectys have a unique mechanism of air exchange in their bodies. The respiratory system of the insects consists of the tracheae that is responsible for the gaseous exchange in and out of tissues. There are spiracles present that guards the trachea. They are valve-like and either remains open, closed, or in a fluttering state.
Explanation of Solution
Intratracheal partial pressure of O2 during the open phase of the spiracles in the tracheae as shown in the figure is 20.4 kPa.
If 1 kPa = 7.5mm Hg, then, Po2 during the open phase will be,7.5*20.4 = 153 mm Hg.
During the closed phase, Po2 decreases to 4–5 kPa.
If 1 kPa = 7.5mm Hg, then, PO2 during the closed phase will be,7.5*5 = 37.5 mm Hg.
During the fluttering phase, if the oxygen concentration is changed as shown in figure B, the PO2 will be calculated as
Case1, If 1 kPa = 7.5mm Hg, 7.5*6.4 = 48 mm Hg.
Case2, If 1 kPa = 7.5mm Hg, 7.5*21.2 = 159 mm Hg.
Case3, If 1 kPa = 7.5mm Hg, 7.5*40.2 = 301.5 mm Hg.
Thus, it can be concluded that the changes in approximate mm Hg of PO2 in a typical open-close fluttering cycle are 153 mm Hg and 7.5 mm Hg during the open and closed phases, respectively. Also, in the tubes of the trachea, in all the three given cases, the pressure was 48 mm Hg, 159 mm Hg, and 301.5 mm Hg, respectively.
Want to see more full solutions like this?
Chapter 48 Solutions
Life: The Science of Biology
- What formula could we use to calculate the rate of respiration in this experiment. Select all that apply: Change in O_2O2 / Change in Time Change in Time / Change in O_2O2 Change in CO_2CO2 / Change in Time Change in Time / Change in O_2O2arrow_forwardHow does one measure Km using a Lineweaver-Burk plot? 1. By determining the reciprocal of the y-intercept 2. By determining the negative reciprocal of the x-intercept 3. By determining the slope and multiplying it by kcat 4. By determining the saturation pointarrow_forwardDetermine the Hill coefficient, n, for myoglobin (Mb), hemoglobin (Hb), and recombinant hemoglobin (HbX). Given the results of the Hill plot and the dissociation curves, which of the following are true? Oxygen binding by HbX is less cooperative than for Hb The Hill coefficient for Hb is equal to the number of hemoglobin O2 binding sites Oxygen binding by Hb is less cooperative than for Mb. HbX will load less O2 in the lungs and unload less O2 in active tissues than normal Hb.arrow_forward
- HbR is reported to have P50=14torr and a Hillcoefficient=1.2. Calculate ΔYO2 for a climber with HbR assuming that, at 14,000 ft (∼4300m), PO2=43mmHg in lungs and PO2=10mmHg in muscle capillaries.arrow_forwardAfter spending a day or more at high altitude (with an oxygen partial pressure of 75 torr), the concentration of 2,3- bisphosphoglycerate (2,3-BPG) in red blood cells increases. What effect would an increased concentration of 2,3-BPG have on the oxygen-binding curve for hemoglobin? Why would this adaptation be beneficial for functioning well at high altitude?arrow_forwardYou want to measure the blood oxygen level in a drop of blood using a pulse oximetry device. If you have a light source at 700nm, and 1mm of path length. What must be the concentration of, a.Oxidized hemoglobin (HbO2) concentration in blood so that the light intensity at the output decreases to the one tenth of input intensity? b. Reduced hemoglobin (Hb) concentration in blood so that the light intensity at the output decreases to the one tenth of input intensity? c.At the concentration you found in b, what is Ii / If for oxidized hemoglobin?arrow_forward
- In the same BP measurement= 120/80, what is the arterial pressure when the left ventricle is at the peak of its contraction? Choices: 120804093arrow_forwardWhy do chymotrypsin and ATCase have different velocity curves?arrow_forwardWith COPD, there is a decreased air flow from the lungs. This coyly be caused by which of the following reasons? Select all that apply.arrow_forward
- If air flows into the lungs with an inlet pressure of 1 bar, how will the air resistance affect the flow, show that using the combined parameter model methodarrow_forwardWhich hemoglobin has a higher affinity for O2 at the tissue pO2 of around 4 kPa?arrow_forwardFor a gradient system with a gradient of 5-90% in 50 min, flow 2 mL/min, column 100 x 4.6 mm i.d., the first peak elutes at 20 min, and the last peak elutes at 50 min. Calculate k* for this system. 2. Can isocratic elution be used for this sample? If so, what is the range of k values to be expected. 3.Propose a way to shorten the gradient run time by eliminating the wait for the first peak to elute at 20 min. 4. For the original conditions of this question, change the conditions necessary to use a 150 x 4.6 mm column while maintaining the same k* value.arrow_forward
 Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning
Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning
