
Concept explainers
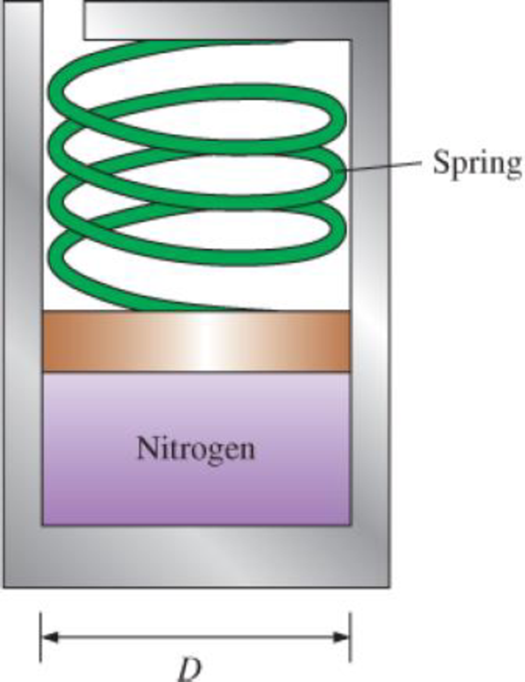
A mass of 10 g of nitrogen is contained in the spring-loaded piston–cylinder device shown in Fig. P4–54. The spring constant is 1 kN/m, and the piston diameter is 10 cm. When the spring exerts no force against the piston, the nitrogen is at 120 kPa and 27°C. The device is now heated until its volume is 10 percent greater than the original volume. Determine the change in the specific internal energy and enthalpy of the nitrogen.
FIGURE P4–54
The change in the internal energy of the nitrogen.
The change in the enthalpy of the nitrogen.
Answer to Problem 54P
The change in the internal energy of the nitrogen is
The change in the enthalpy of the nitrogen is
Explanation of Solution
Write the expression for the initial volume of nitrogen.
Here, the mass of the spring loaded piston cylinder device is
Determine the linear P-v process for spring loaded piston cylinder device.
Here, the system pressure is
Determine the specific heat constant value.
Substitute
From the Equation (IV), the final volume is 10 percent greater than the original volume;
Determine the final temperature of the nitrogen.
Here, the final pressure of the nitrogen is
Determine the internal energy of the spring loaded piston-cylinder device.
Here, the specific heat of constant volume is
Determine the enthalpy of the spring loaded piston-cylinder device.
Here, the specific heat of constant pressure is
Conclusion:
Write the conversion of unit for temperature of 20 C from
Refer Table A-2(a), “Ideal-gas specific heats of various common gases” to obtain the value of gas constant, specific heat of constant volume and pressure for nitrogen gas is
Substitute
Substitute
Substitute
Substitute
Substitute
Thus, the change in the internal energy of the nitrogen is
Substitute
Thus, the change in the enthalpy of the nitrogen is
Want to see more full solutions like this?
Chapter 4 Solutions
Thermodynamics: An Engineering Approach
- A rigid vessel contains contain 5 kg of wet steam at 0.4 MPa. After the addition of 9858 kJ, the steam has a pressure of 0.20 MPa and a temperature of 700C. Determine the initial internal energy and the specific volume of the steam.arrow_forwardA rigid vessel contains 5 kg of wet steam at 0.4 Mpa. After the addition of 9,585 J the steam has a pressure of 2.0 Mpa and a temperature of 700C. Determine the initial internal energy and the specific volume of the steam.arrow_forward1 lbm of carbon dioxide is heated in a constantpressure apparatus. Initially, the carbon dioxide is at 1000 psia and 200°F, and it is heated until its temperature becomes 800°F. Determine the final volume of the carbon dioxide, treating it as (a) an ideal gas and (b) a Benedict-Webb-Rubin gas.arrow_forward
- Steam is contained in a 4-L volume at a pressure of 1.5 MPa and a temperature of 400 C. If the pressure is held constant by expanding the volume while 20 kJ of heat is added, the final temperature is nearestarrow_forwardA piston-cylinder device contains 50 kg of water at 250 kPa and 25°C. The cross-sectional area of the plunger is 0.1 m2. Heat is transferred to the water, causing some of it to evaporate and expand; When the volume reaches 0.2 m3, the piston hits a linear spring whose spring constant is 100 kN/m. More heat is transferred to the water until the plunger rises a further 20 cm. Determine a) the final pressure and temperature and b) the work done during this process. Also show the process on a P-V diagram.arrow_forwardA mass of 10 g of oxygen fill a weighted piston– cylinder device at 20 kPa and 100°C. The device is now cooled until the temperature is 0°C. Determine the change of the volume of the device during this coolingarrow_forward
- A 2 kg mass oxygen expands at constant pressure of 172 kPa in piston-cylinder system from a temperature of 32 oC to a final temperature if 182 oC. Determine the work done.arrow_forwardDetermine the quality of steam at 169.06 kPa when 270 kJ/kg of energy are lost from saturated steam. What is the steam temperature?arrow_forwardThe increase of enthalpy of a certain gas is 141.9 kJ when the pressure increases from 103.448 kPaa to 1034.48 kPaa and the volume decreases from 0.4772 m3 to 0.0745 m3 . Determine (a) the change in internal energy and (b) the final temperature of the gas if the initial temperature is 28 oC.arrow_forward
- 10-kg of R-134a at 300 kPa fills a rigid container whose volume is 14 L. Determine the temperature and total enthalpy in the container. The container is now heated until the pressure is 600 kPa. Determine the temperature and total enthalpy when the heating is completed.arrow_forwardThe radiator of a steam heating system has a volume of 20 L and is filled with superheated water vapor at 200 kPa and 200°C. At this moment both the inlet and the exit valves to the radiator are closed. After a while it is observed that the temperature of the steam drops to 80°C as a result of heat transfer to the room air, which is at 21°C. Assuming the surroundings to be at 0°C, determine the maximum amount of heat that can be supplied to the room if this heat from the radiator is supplied to a heat engine that is driving a heat pump. Assume the heat engine operates between the radiator and the surroundings.arrow_forwardThe radiator of a steam heating system has a volume of 150L and is filled with superheated vapor at 200kPa and 200C. At this moment both the inlet and exit valves to the radiator are closed. Determine the amount of heat that will be transferred to the room when the steam pressure drops to 100 kPaarrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





