
Thermodynamics: An Engineering Approach
9th Edition
ISBN: 9781259822674
Author: Yunus A. Cengel Dr., Michael A. Boles
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4.5, Problem 34P
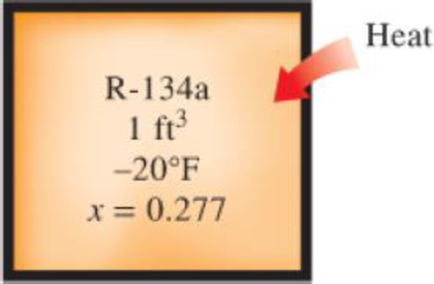
A rigid 1-ft3 vessel contains R-134a originally at −20°F and 27.7 percent quality. The refrigerant is then heated until its temperature is 100°F. Calculate the heat transfer required to do this.
FIGURE P4–34E
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
A kilogram of Argon gas( MW= 40 kg/kmol, k=5/3) is in a piston cylinder set-up. Initially, it has pressure of 30kPa and volume of 0.2m3 and expanded to 20 kPa due to heat transfer. If the process is said to be isovolumic, find initial and final temperatures.
4-68 An insulated piston-cylinder device initially contains 0.3
m³ of carbon dioxide at 200 kPa and 27°C. An electric switch is
turned on, and a 110-V source supplies current to a resistance
heater inside the cylinder for a period of 10 min. The pressure is
held constant during the process, while the volume is doubled.
Determine the current that passes through the resistance heater.
2- A mass of 5 kg of saturated liquid water at 200 kPa is heated at a
constant pressure until the temperature reaches 300°C. Find:
(1) Au, Ah & Av, (2) the work done during this process, (3) the amount of
heat added to the system, and (4) show the process on T-v diagram
Chapter 4 Solutions
Thermodynamics: An Engineering Approach
Ch. 4.5 - Is the boundary work associated with...Ch. 4.5 - On a P-V diagram, what does the area under the...Ch. 4.5 - An ideal gas at a given state expands to a fixed...Ch. 4.5 - Calculate the total work, in kJ, for process 13...Ch. 4.5 - Calculate the total work, in Btu, produced by the...Ch. 4.5 - Nitrogen at an initial state of 300 K, 150 kPa,...Ch. 4.5 - The volume of 1 kg of helium in a pistoncylinder...Ch. 4.5 - A pistoncylinder device with a set of stops...Ch. 4.5 - A mass of 5 kg of saturated water vapor at 150 kPa...Ch. 4.5 - A frictionless pistoncylinder device contains 16...
Ch. 4.5 - 1 m3 of saturated liquid water at 200C is expanded...Ch. 4.5 - Argon is compressed in a polytropic process with n...Ch. 4.5 - A gas is compressed from an initial volume of 0.42...Ch. 4.5 - A mass of 1.5 kg of air at 120 kPa and 24C is...Ch. 4.5 - During some actual expansion and compression...Ch. 4.5 - A frictionless pistoncylinder device contains 5 kg...Ch. 4.5 - During an expansion process, the pressure of a gas...Ch. 4.5 - A pistoncylinder device initially contains 0.4 kg...Ch. 4.5 - A pistoncylinder device contains 0.15 kg of air...Ch. 4.5 - Determine the boundary work done by a gas during...Ch. 4.5 - 1 kg of water that is initially at 90C with a...Ch. 4.5 - An ideal gas undergoes two processes in a...Ch. 4.5 - A pistoncylinder device contains 50 kg of water at...Ch. 4.5 - Prob. 26PCh. 4.5 - A closed system like that shown in Fig. P427E is...Ch. 4.5 - A rigid container equipped with a stirring device...Ch. 4.5 - Complete each line of the following table on the...Ch. 4.5 - A substance is contained in a well-insulated rigid...Ch. 4.5 - A 0.5-m3rigid tank contains refrigerant-134a...Ch. 4.5 - A 20-ft3 rigid tank initially contains saturated...Ch. 4.5 - A rigid 10-L vessel initially contains a mixture...Ch. 4.5 - A rigid 1-ft3 vessel contains R-134a originally at...Ch. 4.5 - A pistoncylinder device contains 5 kg of...Ch. 4.5 - A pistoncylinder device contains 0.5 lbm of water...Ch. 4.5 - 2 kg of saturated liquid water at 150C is heated...Ch. 4.5 - An insulated pistoncylinder device contains 5 L of...Ch. 4.5 - A 40-L electrical radiator containing heating oil...Ch. 4.5 - Steam at 75 kPa and 8 percent quality is contained...Ch. 4.5 - A pistoncylinder device initially contains 0.6 m3...Ch. 4.5 - An insulated tank is divided into two parts by a...Ch. 4.5 - Two tanks (Tank A and Tank B) are separated by a...Ch. 4.5 - Is the energy required to heat air from 295 to 305...Ch. 4.5 - A fixed mass of an ideal gas is heated from 50 to...Ch. 4.5 - A fixed mass of an ideal gas is heated from 50 to...Ch. 4.5 - A fixed mass of an ideal gas is heated from 50 to...Ch. 4.5 - Is the relation u = mcv,avgT restricted to...Ch. 4.5 - Is the relation h = mcp,avgT restricted to...Ch. 4.5 - What is the change in the internal energy, in...Ch. 4.5 - Neon is compressed from 100 kPa and 20C to 500 kPa...Ch. 4.5 - What is the change in the enthalpy, in kJ/kg, of...Ch. 4.5 - A mass of 10 g of nitrogen is contained in the...Ch. 4.5 - Determine the internal energy change u of...Ch. 4.5 - Determine the enthalpy change h of oxygen, in...Ch. 4.5 - Is it possible to compress an ideal gas...Ch. 4.5 - Nitrogen in a rigid vessel is cooled by rejecting...Ch. 4.5 - Nitrogen at 100 psia and 300F in a rigid container...Ch. 4.5 - A pistoncylinder device containing carbon-dioxide...Ch. 4.5 - A 3-m3 rigid tank contains hydrogen at 250 kPa and...Ch. 4.5 - 1 kg of oxygen is heated from 20 to 120C....Ch. 4.5 - A 10-ft3 tank contains oxygen initially at 14.7...Ch. 4.5 - A 4-m 5-m 7-m room is heated by the radiator of...Ch. 4.5 - An insulated rigid tank is divided into two equal...Ch. 4.5 - An ideal gas contained in a pistoncylinder device...Ch. 4.5 - A 4-m 5-m 6-m room is to be heated by a...Ch. 4.5 - An insulated pistoncylinder device initially...Ch. 4.5 - Argon is compressed in a polytropic process with n...Ch. 4.5 - An insulated pistoncylinder device contains 100 L...Ch. 4.5 - Air is contained in a variable-load pistoncylinder...Ch. 4.5 - A mass of 15 kg of air in a pistoncylinder device...Ch. 4.5 - Prob. 73PCh. 4.5 - A pistoncylinder device contains 2.2 kg of...Ch. 4.5 - A pistoncylinder device contains 4 kg of argon at...Ch. 4.5 - A spring-loaded pistoncylinder device contains 5...Ch. 4.5 - Prob. 78PCh. 4.5 - Prob. 79PCh. 4.5 - A 1-kg block of iron is heated from 25 to 75C....Ch. 4.5 - The state of liquid water is changed from 50 psia...Ch. 4.5 - During a picnic on a hot summer day, all the cold...Ch. 4.5 - An ordinary egg can be approximated as a...Ch. 4.5 - Consider a 1000-W iron whose base plate is made of...Ch. 4.5 - Stainless steel ball bearings ( = 8085 kg/m3 and...Ch. 4.5 - In a production facility, 1.6-in-thick 2-ft 2-ft...Ch. 4.5 - Long cylindrical steel rods ( = 7833 kg/m3 and cp...Ch. 4.5 - An electronic device dissipating 25 W has a mass...Ch. 4.5 - Prob. 90PCh. 4.5 - Prob. 91PCh. 4.5 - Is the metabolizable energy content of a food the...Ch. 4.5 - Is the number of prospective occupants an...Ch. 4.5 - Prob. 94PCh. 4.5 - Prob. 95PCh. 4.5 - Prob. 96PCh. 4.5 - Consider two identical 80-kg men who are eating...Ch. 4.5 - A 68-kg woman is planning to bicycle for an hour....Ch. 4.5 - A 90-kg man gives in to temptation and eats an...Ch. 4.5 - A 60-kg man used to have an apple every day after...Ch. 4.5 - Consider a man who has 20 kg of body fat when he...Ch. 4.5 - Consider two identical 50-kg women, Candy and...Ch. 4.5 - Prob. 103PCh. 4.5 - Prob. 104PCh. 4.5 - Prob. 105PCh. 4.5 - Prob. 106PCh. 4.5 - Prob. 107PCh. 4.5 - Prob. 108PCh. 4.5 - Prob. 109RPCh. 4.5 - Prob. 110RPCh. 4.5 - Prob. 111RPCh. 4.5 - Prob. 112RPCh. 4.5 - Prob. 113RPCh. 4.5 - Consider a pistoncylinder device that contains 0.5...Ch. 4.5 - Prob. 115RPCh. 4.5 - Air in the amount of 2 lbm is contained in a...Ch. 4.5 - Air is expanded in a polytropic process with n =...Ch. 4.5 - Nitrogen at 100 kPa and 25C in a rigid vessel is...Ch. 4.5 - Prob. 119RPCh. 4.5 - A mass of 3 kg of saturated liquidvapor mixture of...Ch. 4.5 - A mass of 12 kg of saturated refrigerant-134a...Ch. 4.5 - Prob. 122RPCh. 4.5 - A pistoncylinder device contains helium gas...Ch. 4.5 - Prob. 124RPCh. 4.5 - Prob. 125RPCh. 4.5 - Prob. 126RPCh. 4.5 - Prob. 127RPCh. 4.5 - Water is boiled at sea level in a coffeemaker...Ch. 4.5 - The energy content of a certain food is to be...Ch. 4.5 - Prob. 130RPCh. 4.5 - An insulated pistoncylinder device initially...Ch. 4.5 - An insulated rigid tank initially contains 1.4 kg...Ch. 4.5 - In order to cool 1 ton of water at 20C in an...Ch. 4.5 - A 0.3-L glass of water at 20C is to be cooled with...Ch. 4.5 - A well-insulated 3-m 4m 6-m room initially at 7C...Ch. 4.5 - Prob. 137RPCh. 4.5 - Prob. 138RPCh. 4.5 - Prob. 140RPCh. 4.5 - A pistoncylinder device initially contains 0.35 kg...Ch. 4.5 - Two 10-ft3 adiabatic tanks are connected by a...Ch. 4.5 - Prob. 143RPCh. 4.5 - Prob. 144RPCh. 4.5 - A 3-m3 rigid tank contains nitrogen gas at 500 kPa...Ch. 4.5 - A 0.5-m3 rigid tank contains nitrogen gas at 600...Ch. 4.5 - A well-sealed room contains 60 kg of air at 200...Ch. 4.5 - A room contains 75 kg of air at 100 kPa and 15C....Ch. 4.5 - Prob. 149FEPCh. 4.5 - A pistoncylinder device contains 5 kg of air at...Ch. 4.5 - Prob. 151FEPCh. 4.5 - A 2-kW electric resistance heater submerged in 5...Ch. 4.5 - Prob. 153FEPCh. 4.5 - 1.5 kg of liquid water initially at 12C is to be...Ch. 4.5 - Prob. 155FEPCh. 4.5 - An ordinary egg with a mass of 0.1 kg and a...Ch. 4.5 - Prob. 157FEPCh. 4.5 - A 6-pack of canned drinks is to be cooled from 18C...Ch. 4.5 - Prob. 159FEPCh. 4.5 - An ideal gas has a gas constant R = 0.3 kJ/kgK and...Ch. 4.5 - A pistoncylinder device contains an ideal gas. The...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Similar questions
- 0.5 kg of steam at 250°C is enclosed behind a piston in a cylinder at 0.9MPa. with the piston rigidly held in position, the cylinder is cooled until the pressure is 0.7MPa. Find the condition of the steam at the lower pressure. Calculate the change in internal energy and explain why this value represent the quantity of heat transfer during the cooling process. If the amount of heat is now transferred back into steam while the pressure remain constant at 0.7MPa, determine the final condition of the steam. Take the specific heat of superheated steam as 2kJ/kg K. (0.952, -115.7kJ, superheat temperature 231.1°C) 14. 15. A steam turbine is developed 3000kW from steam supplied at a pressure of 3 MPa and 300°C. The exhaust pressure is 7 kPa and the condition at exhaust is 0.9 dry. Determine a-the required steam flow rate in kg/s b-the cross sectional area of the turbine exhaust if the steam velocity is 75 m/s. (4.52 kg/s, 1.114 m²) initiall 1:arrow_forward10 lbm of ammonia (MW = 17), is heated inside a cylinder equipped with a piston. The initial pressure and temperature is 63 psi and 50°F. If 500 BTU of heat was added at a constant pressure until the temperature doubled, what is the work done by the gas in BTU? Also draw and label the P-V and T-S diagram.arrow_forwardSteam is contained in a 2-L volume at a pressure of 2.0 MPa and a temperature of 400°C. If the pressure is held constant by expanding the volume while 20 kJ of heat is added, what is the final temperature and the work done?arrow_forward
- 4-65E A 10 ft³ tank contains oxygen initially at 14.7 psia and 80°F. A paddle wheel within the tank is rotated until the pressure inside rises to 20 psia. During the process 20 Btu of heat is lost to the surroundings. Determine the paddle-wheel work done. Neglect the energy stored in the paddle wheel.arrow_forward10 lbm of ammonia (MW = 17), is heated inside a cylinder equipped with a piston. The initial pressure and temperature is 63 psi and 50°F. If 500 BTU of heat was added at a constant pressure until the temperature doubled, what is the work done by the gas in BTU? Draw and label the P-V and T-S diagram.arrow_forward4-118 A piston-cylinder device contains helium gas initially at 100 kPa, 10°C, and 0.2 m³. The helium is now compressed in a polytropic process (PV=constant) to 700 kPa and 290°C. Determine the heat loss or gain during this process.arrow_forward
- T=119°℃ 2 A dividing wall divides an insulated container into two compartments as shown in the figure. Compartment A contains 0.3 kg of water at T (°C) and 10 MPa pressure. Compartment B contains 10 kg of water at T (°C) and 0.1 MPa pressure. The dividing wall is removed and water in both compartments fills the entire container. If the final temperature is T (C), determine the final pressure and the heat transfer involved in the mixing process B Aarrow_forwardA piston/cylinder assembly with a spring contains 0.075 kg of water. The piston is initially resting on the stops and the water has a temperature of T1 = 80°C and a quality of x1 = 30%. Heat is added to the water causing the piston to lift off the stops, rise up and compress the spring. Heating contin- ues until a final state is reached and the pressure is 200 kPa. The vertical distance between the top of the piston when it is resting on the stops and the bottom of the spring is Ay = 0.6 m, as shown in the figure. Assume the piston is massless (m, = 0), the area of the piston is A, = 0.12 m², the spring constant is k, = 40 kN/m and atmospheric pressure is Po = 100 kPa. a) Determine the volume, pressure and temperature of the water when the piston just lifts off the stops, V2, P2 and T2. b) Determine the pressure and specific volume of the water when the piston just touches but is not yet compressing the spring, P3 and v3. c) Determine the final volume of water in the piston/cylinder…arrow_forward4-40 Steam at 75 kPa and 8 percent quality is contained in a spring-loaded piston-cylinder device, as shown in Fig. P4-40. with an initial volume of 2 m³. Steam is now heated until its volume is 5 m³ and its pressure is 225 kPa. Determine the heat transferred to and the work produced by the steam during this process. FIGURE P4-40 www Steamarrow_forward
- (a) In the thermodynamic laboratory, student Ali prepared a piston-cylinder device which contains refrigerant-134a at 1000 kPa and 50°C. The mass of the refrigerant is 6 kg. The next step of the experiment involves cooling the refrigerant at constant pressure until its state become liquid at 24°C. Show the process on T-v diagram and determine the heat loss from the system. (b) A heat exchanger as shown in Figure 1 is used to cool water flowing in at 1 kg/s water, 10 kPa and quality 0.95 to saturated liquid at 10 kPa. The cooling fluid is water taken from a lake at 20°C and it exit the heat exchanger at 30°C. Calculate the flow rate of cooling water. Assume that the heat exchange is well insulated and there is no heat loss. Lake water Figure 1: Heat exchangerarrow_forwardWater initially at 200 kPa and 300°C is contained in a piston-cylinder device fitted with stops. The water is allowed to cool at constant pressure until it exists as a saturated vapor and the piston rests on the stops. Then the water continues to cool until the pressure is 100 kPa. Water 200 kPa 300°C Find the overall change in internal energy between the initial and final states per unit mass of water. Use data from the tables. (You must provide an answer before moving on to the next part.) The overall change in internal energy is K kJ/kg.arrow_forward3- A mass of 10 kg of saturated refrigerant-134a vapor is contained in a piston-cylinder device at 600 kPa. Now 260 kJ of heat is transferred to the refrigerant at constant pressure while a 110-V source supplies current to a resistor within the cylinder for 30 min. If the final temperature is 80°C, the electric current supplied to the resistor is: (a) 1.173A (b) 1.273A (c) 1.373A (d) 1.473A (e) 1.573Aarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY

Elements Of Electromagnetics
Mechanical Engineering
ISBN:9780190698614
Author:Sadiku, Matthew N. O.
Publisher:Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:9780134319650
Author:Russell C. Hibbeler
Publisher:PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:9781259822674
Author:Yunus A. Cengel Dr., Michael A. Boles
Publisher:McGraw-Hill Education

Control Systems Engineering
Mechanical Engineering
ISBN:9781118170519
Author:Norman S. Nise
Publisher:WILEY

Mechanics of Materials (MindTap Course List)
Mechanical Engineering
ISBN:9781337093347
Author:Barry J. Goodno, James M. Gere
Publisher:Cengage Learning

Engineering Mechanics: Statics
Mechanical Engineering
ISBN:9781118807330
Author:James L. Meriam, L. G. Kraige, J. N. Bolton
Publisher:WILEY
First Law of Thermodynamics, Basic Introduction - Internal Energy, Heat and Work - Chemistry; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=NyOYW07-L5g;License: Standard youtube license