
Concept explainers
A mass of 3 kg of saturated liquid–vapor mixture of water is contained in a piston–cylinder device at 160 kPa. Initially, 1 kg of the water is in the liquid phase and the rest is in the vapor phase. Heat is now transferred to the water, and the piston, which is resting on a set of stops, starts moving when the pressure inside reaches 500 kPa. Heat transfer continues until the total volume increases by 20 percent. Determine (a) the initial and final temperatures, (b) the mass of liquid water when the piston first starts moving, and (c) the work done during this process. Also, show the process on a P-v diagram.
FIGURE P4–120
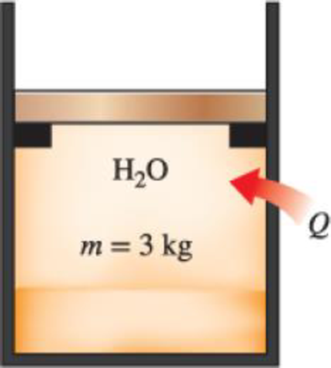
(a)
The initial temperature of the piston cylinder device.
The final temperature of the piston cylinder device.
Answer to Problem 120RP
The initial temperature of the piston cylinder device is
The final temperature of the piston cylinder device is
Explanation of Solution
Determine the total initial volume of piston cylinder device.
Here, the mass of the liquid phase is
Determine the total volume of the piston cylinder device at final state.
Determine the specific volume of the piston cylinder device at final state.
Here, the mass of the saturated liquid vapour mixture of water is contained in a piston cylinder device is
Conclusion:
Write the formula of interpolation method of two variables.
Here, the variables denote by x and y is saturated pressure and saturated temperature.
For initial temperature of the piston cylinder device.
Show the temperature at pressure of 150 kPa, 160 kPa, and 175 kPa as in Table (1).
|
Pressure, kPa |
Temperature, C |
| 150 kPa | 111.35 |
| 160 kPa | |
| 175 kPa | 116.04 |
Substitute the value of x and y from Table (1) in Equation (IV) to calculate the value of initial temperature
Thus, the initial temperature of the piston cylinder device is
For specific volume of saturated liquid of the piston cylinder device.
Show the specific volume of saturated liquid at pressure of 150 kPa, 160 kPa, and 175 kPa as in Table (2).
|
Pressure, kPa |
Specific volume of saturated liquid, |
| 150 kPa | 0.001053 |
| 160 kPa | |
| 175 kPa | 0.001057 |
Substitute the value of x and y from Table (2) in Equation (IV) to calculate the value of specific volume of saturated liquid
For specific volume of saturated vapour of the piston cylinder device.
Show the specific volume of saturated vapour at pressure of 150 kPa, 160 kPa, and 175 kPa as in Table (3).
|
Pressure, kPa |
Specific volume of saturated vapour, |
| 150 kPa | 1.1594 |
| 160 kPa | |
| 175 kPa | 1.0037 |
Substitute the value of x and y from Table (3) in Equation (IV) to calculate the value of specific volume of saturated vapour
Substitute
Substitute
Substitute
The unit conversion of pressure from kPa to MPa.
For temperature of the piston cylinder device at final state.
Show the temperature at specific volume of the piston cylinder device at final state at
|
specific volume of the piston cylinder device at final state, |
Temperature, |
| 600 | |
| 700 |
Substitute the value of x and y from Table (4) in Equation (IV) to calculate the value of temperature of the piston cylinder device at final state
Thus, the final temperature of the piston cylinder device is
(b)
The mass of liquid water when the piston first starts moving.
Answer to Problem 120RP
The mass of liquid water when the piston first starts moving is
Explanation of Solution
Determine the specific volume of the piston cylinder device at this state.
Here, the mass of the saturated liquid vapour mixture of water is contained in a piston cylinder device is
Conclusion:
Since,
Substitute
Therefore, the value of specific volume of the piston cylinder device at this state is greater than
Thus, the mass of liquid water when the piston first starts moving is
(c)
The work done during the process state 2 and 3.
Answer to Problem 120RP
The work done during the process state 2 and 3 is
Explanation of Solution
Determine the work done in constant pressure process.
Conclusion:
Substitute
Thus, the work done during the process state 2 and 3 is
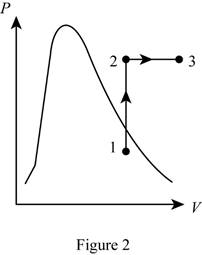
Show the P-v diagram of this process.
Want to see more full solutions like this?
Chapter 4 Solutions
Thermodynamics: An Engineering Approach
- A 4-L pressure cooker has an operating pressure of 175 kPa. Initially, one-half of the volume is filled with liquid water and the other half by water vapor. The cooker is now placed on top of a 750-W electrical heating unit that is kept on for 20 min. Assuming the surroundings to be at 25°C and 100 kPa, determine the amount of water that remained in the cooker.arrow_forwardWater is contained in a constant volume tank as a saturated mixture at 90 degrees C. When it is heated, the water pressure and temperature become 200kPa and 300 degrees C. Determine the initail quality of the water in the tank, the heat transfer per unit mass of the water, and the change in entropy per unit mass of the water.arrow_forwardWhat is the maximum volume that 3 kg of oxygen at 950 kPa and 373°C can be adiabatically expanded to in a piston–cylinder device if the final pressure is to be 100 kPa?arrow_forward
- A constant-pressure R-134a vapor separation unit separates the liquid and vapor portions of a saturated mixture into two separate outlet streams. Determine the flow power needed to pass 6 L/s of R-134a at 320 kPa and 55 percent quality through this unit. What is the mass flow rate, in kg/s, of the two outlet streams?arrow_forwardLiquid water at 200 kPa and 15°C is heated in a chamber by mixing it with superheated steam at 200 kPa and 200°C. Liquid water enters the mixing chamber at a rate of 4 kg/s, and the chamber is estimated to lose heat to the surrounding air at 25°C at a rate of 600 kJ/min. If the mixture leaves the mixing chamber at 200 kPa and 80°C, determine the mass flow rate of the superheated steam.arrow_forwardDetermine the enthalpy of liquid water at 100°C and 15 MPa (a) by using compressed liquid tables, (b) by approximating it as a saturated liquid, and (c) by using the correction givenarrow_forward
- Determine the quality of steam at 169.06 kPa when 270 kJ/kg of energy are lost from saturated steam. What is the steam temperature?arrow_forwardA room is filled with saturated steam at 100°C with enthalpy of 2675.6 kJ/kg. Now a 5-kg bowling ball at 25°C is brought to the room. Heat is transferred to the ball from the steam, and the temperature of the ball rises to 100°C while some steam condenses on the ball as it loses heat (but it still remains at 100°C). The specific heat of the ball can be taken to be 1.8 kJ/kg · C. The approximate mass of steam that condensed during this process is? what is the answer? a. 80 g b. 252 g c. 351 g d. 128 garrow_forwardA room is filled with saturated steam at 100°C with enthalpy of 2675.6 kJ/kg. Now a 5-kg bowling ball at 25°C is brought to the room. Heat is transferred to the ball from the steam, and the temperature of the ball rises to 100°C while some steam condenses on the ball as it loses heat (but it still remains at 100°C). The specific heat of the ball can be taken to be 1.8 kJ/kg · C. The approximate mass of steam that condensed during this process isarrow_forward
- A 240-m3 rigid tank is filled with a saturated liquid– vapor mixture of water at 200 kPa. If 25 percent of the mass is liquid and 75 percent of the mass is vapor, the total mass in the tank is (a) 240 kg (b) 265 kg (c) 307 kg (d) 361 kg (e) 450 kgarrow_forward25 kg of refrigerant 134a, at 300 kPa, fill a rigid container whose volume is 30 L. Determine the temperature and total enthalpy in the container. Now the container is heated, until the pressure is 700 kPa. Determine the temperature and the total enthalpy of the refrigerant when heating is complete.arrow_forwardArgon is compressed in a polytropic process with n = 1.2 from 120 kPa and 30°C to 1200 kPa in a piston– cylinder device. Determine the final temperature of the argon.arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





