
Chemistry: Structure and Properties (2nd Edition)
2nd Edition
ISBN: 9780134293936
Author: Nivaldo J. Tro
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4, Problem 92E
Write a ratio showing the relationship between the molar amounts of each element for each compound.
a.

b.
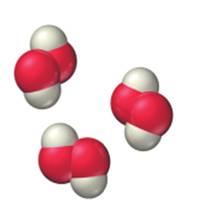
c.
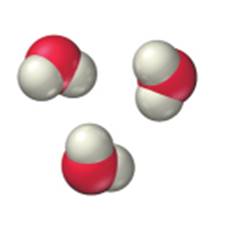
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
How many grams of C are combined with 3.75 ✕ 1023 atoms of H in the compound C5H12?
e.
f. CH3O.
יון
Br
NaOCH3
OCH 3
Br
H₂O
Shkruani me fjale dhe me formula kimike reaksionin e bashkeveprimit te natriumit me ujin
Chapter 4 Solutions
Chemistry: Structure and Properties (2nd Edition)
Ch. 4 - How do the properties of compounds compare to the...Ch. 4 - What is a chemical bond? Why do chemical bonds...Ch. 4 - Explain the difference between an ionic bond and a...Ch. 4 - List and describe the different ways to represent...Ch. 4 - What is the difference between an empirical...Ch. 4 - Prob. 6ECh. 4 - Prob. 7ECh. 4 - Prob. 8ECh. 4 - How can you use Lewis structures to determine the...Ch. 4 - What is lattice energy?
Ch. 4 - Why is the formation of solid sodium chloride from...Ch. 4 - Prob. 12ECh. 4 - Prob. 13ECh. 4 - Prob. 14ECh. 4 - Prob. 15ECh. 4 - Prob. 16ECh. 4 - Prob. 17ECh. 4 - How does the Lewis model for covalent bonding...Ch. 4 - Explain howto nans molecular inorganic compounds.Ch. 4 - Prob. 20ECh. 4 - What is the formula mass for a compound? Why is it...Ch. 4 - Explain how the information in a chemical formula...Ch. 4 - What is mass percent composition? Why is it...Ch. 4 - Which kinds of conversion factors are inherent in...Ch. 4 - Which kind of chemical formula can be obtained...Ch. 4 - Prob. 26ECh. 4 - Prob. 27ECh. 4 - Prob. 28ECh. 4 - Prob. 29ECh. 4 - Prob. 30ECh. 4 - Prob. 31ECh. 4 - Determine the empirical formula for the compound...Ch. 4 - Determine the number of each type of atom in each...Ch. 4 - Determine the number of each type of atom in each...Ch. 4 - Write a chemical formula for each molecular model....Ch. 4 - Prob. 36ECh. 4 - Prob. 37ECh. 4 - Write an electron configuration for Ne. Then write...Ch. 4 - Prob. 39ECh. 4 - Write a Lewis symbol for each atom or ion. a. S2-...Ch. 4 - Prob. 41ECh. 4 - Write the Lewis symbols that represent the ions in...Ch. 4 - Prob. 43ECh. 4 - Prob. 44ECh. 4 - The lattice energy of CsF is -744 kJ/mol, whereas...Ch. 4 - Rubidium iodide has a lattice energy of-617...Ch. 4 - Prob. 47ECh. 4 - Prob. 48ECh. 4 - Prob. 49ECh. 4 - Prob. 50ECh. 4 - Prob. 51ECh. 4 - Prob. 52ECh. 4 - Prob. 53ECh. 4 - Prob. 54ECh. 4 - Prob. 55ECh. 4 - Prob. 56ECh. 4 - Prob. 57ECh. 4 - Prob. 58ECh. 4 - Prob. 59ECh. 4 - Prob. 60ECh. 4 - Use covalent Lewis structures to explain why each...Ch. 4 - Use covalent Lewis structures to explain why the...Ch. 4 - Prob. 63ECh. 4 - Prob. 64ECh. 4 - Prob. 65ECh. 4 - Prob. 66ECh. 4 - Name each compound. (Refer to the nomenclature...Ch. 4 - Prob. 68ECh. 4 - Prob. 69ECh. 4 - Prob. 70ECh. 4 - Calculate the formula mass for each compound. NO2...Ch. 4 - Prob. 72ECh. 4 - Calculate the number of moles in each sample 72.5...Ch. 4 - Calculate the mass of each sample 15.7 mol HNO3...Ch. 4 - Determine the number of moles (of molecules or...Ch. 4 - Determine the number of moles (of molecules or...Ch. 4 - How many molecules are in each sample? 6.5 g H2O...Ch. 4 - Prob. 78ECh. 4 - Calculate the mass (in g) of each sample. 5.94 x...Ch. 4 - Calculate the mass (in g) of each sample 4.5 x...Ch. 4 - A sugar crystal contains approximately 1.8 x 1017...Ch. 4 - A salt crystal has a mass of 0.12 mg. How many...Ch. 4 - Calculate the mass percent composition of carbon...Ch. 4 - Calculate the mass percent composition of nitrogen...Ch. 4 - Most fertilizers consist of nitrogen-containing...Ch. 4 - Iron in the earth is in the form of iron ore....Ch. 4 - Copper(ll) fluoride contains 37.42% F by mass....Ch. 4 - Silver chloride, often used in silver plating,...Ch. 4 - The iodide ion is a dietary mineral essential to...Ch. 4 - The American Dental Association recommends that an...Ch. 4 - Write a ratio showing the relationship between the...Ch. 4 - Write a ratio showing the relationship between the...Ch. 4 - Determine the number of moles of hydrogen atoms in...Ch. 4 - Determine the number of moles of oxygen atoms in...Ch. 4 - Calculate mass (in grams) of sodium in 8.5 g of...Ch. 4 - Calculate the mass (in kilograms) of chlorine in...Ch. 4 - A chemist decomposes samples of several compounds;...Ch. 4 - A chemist decomposes samples of several compounds;...Ch. 4 - Calculate the empirical formula for each stimulant...Ch. 4 - Calculate the empirical formula for each natural...Ch. 4 - The elemental mass percent composition of...Ch. 4 - The elemental mass percent composition of ascorbic...Ch. 4 - A 0.77-mg sample of nitrogen reacts with chlorine...Ch. 4 - A 45.2-mg sample of phosphorus reacts with...Ch. 4 - The empirical formula and molar mass of several...Ch. 4 - The malar mass and empirical formula of several...Ch. 4 - Combustion analysis of a hydrocarbon produced...Ch. 4 - Combustion analysis of naphthalene, a hydrocarbon...Ch. 4 - The foul odor of rancid butter is due largely to...Ch. 4 - Tartaric acid is the white, powdery substance that...Ch. 4 - Prob. 111ECh. 4 - Prob. 112ECh. 4 - Prob. 113ECh. 4 - Prob. 114ECh. 4 - How many molecules of ethanol (C2H5OH) (the...Ch. 4 - A drop of water has a volume of approximately 0.05...Ch. 4 - Determine the chemical formula of each compound...Ch. 4 - Determine the chemical formula of each compound...Ch. 4 - A Freon™ leak in the air conditioning system of an...Ch. 4 - Prob. 120ECh. 4 - A metal (M) forms a compound with the formula...Ch. 4 - A metal (M) forms an oxide with the formula M2O....Ch. 4 - Estradiol is a female sexual hormone that causes...Ch. 4 - Fructose is a common sugar found in fruit....Ch. 4 - Combustion analysis of a 13.42-g sample of equilin...Ch. 4 - Prob. 126ECh. 4 - Epsom salts is a hydrated ionic compound with the...Ch. 4 - A hydrate of copper(ll) chloride has the following...Ch. 4 - A compound of molar mass 177 g/mol contains only...Ch. 4 - Researchers obtain the following data from...Ch. 4 - Find the total number of atoms in a sample of...Ch. 4 - Vanadium forms four different oxides in which the...Ch. 4 - The chloride of an unknown metal is believed to...Ch. 4 - Prob. 134ECh. 4 - A chromium-containing compound has the formula...Ch. 4 - Prob. 136ECh. 4 - Prob. 137ECh. 4 - Prob. 138ECh. 4 - A mixture of NaCI and NaBr has a mass of 2.00 g...Ch. 4 - Three pure compounds form when 1.00-g samples of...Ch. 4 - A mixture of CaCO3 and (NH4)2CO3is 61.9% CO3 by...Ch. 4 - A mixture of 50.0 g of S and 1.00 x 102 g of CI2...Ch. 4 - Because of increasing evidence of damage to the...Ch. 4 - A particular coal contains 2.55% sulfur by mass....Ch. 4 - Lead is found in Earth’s crust as several...Ch. 4 - A 2.52-g sample of a compound containing only...Ch. 4 - Prob. 147ECh. 4 - The elements X and Y form a compound that is 40% X...Ch. 4 - A compound of X and Y is 13 X by mass. The atomic...Ch. 4 - A mixture of carbon and sulfur has a mass of 9.0...Ch. 4 - When molecules are represented by molecular...Ch. 4 - Prob. 152ECh. 4 - Explain the problem with this statement and...Ch. 4 - Without doing any calculations, arrange the...Ch. 4 - Prob. 155ECh. 4 - Prob. 156ECh. 4 - Prob. 157ECh. 4 - A compound isolated from the rind of lemons is...Ch. 4 - Prob. 159ECh. 4 - What is the empirical formula of the compound with...Ch. 4 - Which substance is an ionic compound? He N2O4 CCl4...Ch. 4 - Prob. 3SAQCh. 4 - Prob. 4SAQCh. 4 - Prob. 5SAQCh. 4 - Prob. 6SAQCh. 4 - Prob. 7SAQCh. 4 - What is the correct Lewis symbol for S?Ch. 4 - How many CH2Cl2 molecules are there in 25.0 g of...Ch. 4 - List the elements in the compound CF2Cl2 in order...Ch. 4 - Determine the mass of potassium in 35.5 g of KBr....Ch. 4 - A compound is 52.14% C, 13.13% H, and 34.73% O by...Ch. 4 - A compound has the empirical formula CH2O and a...Ch. 4 - Combustion of 30.42 g of a compound containing...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Don't used Ai solutionarrow_forward5. A solution of sucrose is fermented in a vessel until the evolution of CO2 ceases. Then, the product solution is analyzed and found to contain, 45% ethanol; 5% acetic acid; and 15% glycerin by weight. If the original charge is 500 kg, evaluate; e. The ratio of sucrose to water in the original charge (wt/wt). f. Moles of CO2 evolved. g. Maximum possible amount of ethanol that could be formed. h. Conversion efficiency. i. Per cent excess of excess reactant. Reactions: Inversion reaction: C12H22O11 + H2O →2C6H12O6 Fermentation reaction: C6H12O6 →→2C2H5OH + 2CO2 Formation of acetic acid and glycerin: C6H12O6 + C2H5OH + H₂O→ CH3COOH + 2C3H8O3arrow_forwardShow work. don't give Ai generated solution. How many carbons and hydrogens are in the structure?arrow_forward
- 13. (11pts total) Consider the arrows pointing at three different carbon-carbon bonds in the molecule depicted below. Bond B 2°C. +2°C. cleavage Bond A •CH3 + 26.← Cleavage 2°C. + Bond C +3°C• CH3 2C Cleavage E 2°C. 26. weakest bond Intact molecule Strongest 3°C 20. Gund Largest argest a. (2pts) Which bond between A-C is weakest? Which is strongest? Place answers in appropriate boxes. C Weakest bond A Produces Most Bond Strongest Bond Strongest Gund produces least stable radicals Weakest Stable radical b. (4pts) Consider the relative stability of all cleavage products that form when bonds A, B, AND C are homolytically cleaved/broken. Hint: cleavage products of bonds A, B, and C are all carbon radicals. i. Which ONE cleavage product is the most stable? A condensed or bond line representation is fine. 13°C. formed in bound C cleavage ii. Which ONE cleavage product is the least stable? A condensed or bond line representation is fine. • CH3 methyl radical Formed in Gund A Cleavage c.…arrow_forwardBr. COOH Br, FCH COOH E FeBr ASOCI B NH (CH,CO),OD Br₂ 2 C alcKOHarrow_forwardFind A to F (all)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning
Step by Step Stoichiometry Practice Problems | How to Pass ChemistryMole Conversions Made Easy: How to Convert Between Grams and Moles; Author: Ketzbook;https://www.youtube.com/watch?v=b2raanVWU6c;License: Standard YouTube License, CC-BY