
Each of the following ground-state electron configurations represents one or more of the transition metal ions in Figure 4.12. Identify the ion or ions represented by each.

(a)
Interpretation: The ion or ions of transition metals that is represented by the given ground state electronic configuration has to be identified.
Concept Introduction:
- The d-block elements in the periodic table are known as transition metals. When looking at the first series of d-block elements the 4s orbital is filled first before the filling of 3d orbital happens. Therefore, when electron is removed from d-block elements, the two electrons from the “s” orbital is removed first followed by the “d” orbital. This is the reason why many of the transition metals form ions in “+2” state. Two possible oxidation states are there for transition metals namely “+2” and “+3”.
- For simpler representation of ions or atoms, the electronic configuration of the completed octet noble gas configuration is considered and the remaining orbital alone is shown explicitly. The ground-state configuration of the noble gases are given below,
To identify: The transition metal ion or ions that represent the given ground-state electronic configuration.
Answer to Problem 4.85QP
Answer
The transition metal ion found for (a) is
Explanation of Solution
Ground-state electronic configuration of the given ion (a) is,
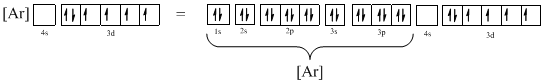
The atomic ground-state configuration is given in the problem statement. From this we can find that the given ion has an empty 4s orbital and 3d sub-shell that has six electrons.
Identifying the ions as follows,
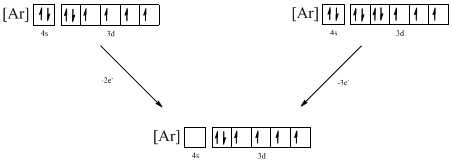
From the configuration given in the problem statement, we can conclude that the ion is formed by removing two electrons from 4s orbital. Another ion can be formed by removing electron from 4s orbital and 3d orbital. Therefore there are two possibility for the ions to have the given electron configuration. It is found to be
(b)
Interpretation: The ion or ions of transition metals that is represented by the given ground state electronic configuration has to be identified.
Concept Introduction:
- The d-block elements in the periodic table are known as transition metals. When looking at the first series of d-block elements the 4s orbital is filled first before the filling of 3d orbital happens. Therefore, when electron is removed from d-block elements, the two electrons from the “s” orbital is removed first followed by the “d” orbital. This is the reason why many of the transition metals form ions in “+2” state. Two possible oxidation states are there for transition metals namely “+2” and “+3”.
- For simpler representation of ions or atoms, the electronic configuration of the completed octet noble gas configuration is considered and the remaining orbital alone is shown explicitly. The ground-state configuration of the noble gases are given below,
To identify: The transition metal ion or ions that represent the given ground-state electronic configuration.
Answer to Problem 4.85QP
Answer
The transition metal ion found for (b) is
Explanation of Solution
Ground-state electronic configuration of the given ion (b) is,

The atomic ground-state configuration is given in the problem statement. From this we can find that the given ion has an empty 5s orbital and 4d sub-shell that has ten electrons.
Identifying the ion as follows,
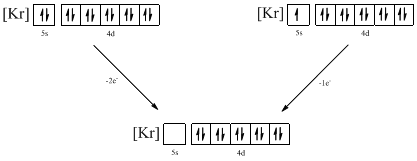
From the configuration given in the problem statement, we can conclude that the ion is formed by removing two electrons from 5s orbital. Another possibility is that the 5s orbital may be singly filled. Therefore, there are two possibility of ions to have the given ground-state electronic configuration. It is found to be
(c)
Interpretation: The ion or ions of transition metals that is represented by the given ground state electronic configuration has to be identified.
Concept Introduction:
- The d-block elements in the periodic table are known as transition metals. When looking at the first series of d-block elements the 4s orbital is filled first before the filling of 3d orbital happens. Therefore, when electron is removed from d-block elements, the two electrons from the “s” orbital is removed first followed by the “d” orbital. This is the reason why many of the transition metals form ions in “+2” state. Two possible oxidation states are there for transition metals namely “+2” and “+3”.
- For simpler representation of ions or atoms, the electronic configuration of the completed octet noble gas configuration is considered and the remaining orbital alone is shown explicitly. The ground-state configuration of the noble gases are given below,
To identify: The transition metal ion or ions that represent the given ground-state electronic configuration.
Answer to Problem 4.85QP
Answer
The transition metal ion found for (c) is
Explanation of Solution
Ground-state electronic configuration of the given ion (c) is,
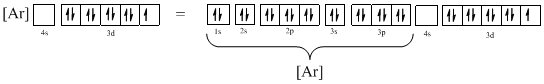
The atomic ground-state configuration is given in the problem statement. From this we can find that the given ion has an empty 4s orbital and 3d sub-shell that has nine electrons.
Identifying the ion as follows,
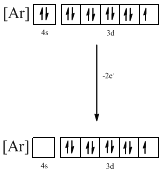
From the configuration given in the problem statement, we can conclude that the ion is formed by removing one electron from 4s orbital and one electron form 3d orbital. The ion that has the given ground-state electronic configuration is found to be
(d)
Interpretation: The ion or ions of transition metals that is represented by the given ground state electronic configuration has to be identified.
Concept Introduction:
- The d-block elements in the periodic table are known as transition metals. When looking at the first series of d-block elements the 4s orbital is filled first before the filling of 3d orbital happens. Therefore, when electron is removed from d-block elements, the two electrons from the “s” orbital is removed first followed by the “d” orbital. This is the reason why many of the transition metals form ions in “+2” state. Two possible oxidation states are there for transition metals namely “+2” and “+3”.
- For simpler representation of ions or atoms, the electronic configuration of the completed octet noble gas configuration is considered and the remaining orbital alone is shown explicitly. The ground-state configuration of the noble gases are given below,
To identify: The transition metal ion or ions that represent the given ground-state electronic configuration.
Answer to Problem 4.85QP
Answer
The transition metal ion found for (d) is
Explanation of Solution
Ground-state electronic configuration of the given ion (d) is,
The atomic ground-state configuration is given in the problem statement. From this we can find that the given ion has an empty 4s orbital and 3d sub-shell that has ten electrons.
Identifying the ion as follows,
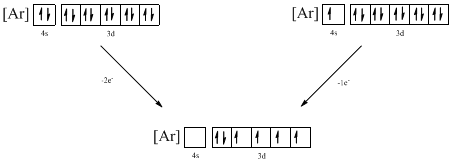
From the configuration given in the problem statement, we can conclude that the ion is formed by removing one electron from 4s orbital. Another possibility is that two electrons can be removed from the 4s orbital. Therefore, there are two possibility of ions to have the given ground-state electronic configuration. It is found to be
(e)
Interpretation: The ion or ions of transition metals that is represented by the given ground state electronic configuration has to be identified.
Concept Introduction:
- The d-block elements in the periodic table are known as transition metals. When looking at the first series of d-block elements the 4s orbital is filled first before the filling of 3d orbital happens. Therefore, when electron is removed from d-block elements, the two electrons from the “s” orbital is removed first followed by the “d” orbital. This is the reason why many of the transition metals form ions in “+2” state. Two possible oxidation states are there for transition metals namely “+2” and “+3”.
- For simpler representation of ions or atoms, the electronic configuration of the completed octet noble gas configuration is considered and the remaining orbital alone is shown explicitly. The ground-state configuration of the noble gases are given below,
To identify: The transition metal ion or ions that represent the given ground-state electronic configuration.
Answer to Problem 4.85QP
Answer
The transition metal ion found for (e) is
Explanation of Solution
Ground-state electronic configuration of the given ion (e) is,
The atomic ground-state configuration is given in the problem statement. From this we can find that the given ion has an empty 4s orbital and 3d sub-shell that has three electrons.
Identifying the ion as follows,
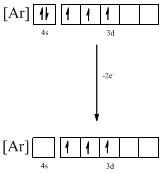
From the configuration given in the problem statement, we can conclude that the ion is formed by removing two electrons from 4s orbital. Therefore, there is only one possibility for the given ground-state electronic configuration. The ion is found to be
(f)
Interpretation: The ion or ions of transition metals that is represented by the given ground state electronic configuration has to be identified.
Concept Introduction:
- The d-block elements in the periodic table are known as transition metals. When looking at the first series of d-block elements the 4s orbital is filled first before the filling of 3d orbital happens. Therefore, when electron is removed from d-block elements, the two electrons from the “s” orbital is removed first followed by the “d” orbital. This is the reason why many of the transition metals form ions in “+2” state. Two possible oxidation states are there for transition metals namely “+2” and “+3”.
- For simpler representation of ions or atoms, the electronic configuration of the completed octet noble gas configuration is considered and the remaining orbital alone is shown explicitly. The ground-state configuration of the noble gases are given below,
To identify: The transition metal ion or ions that represent the given ground-state electronic configuration.
Answer to Problem 4.85QP
Answer
The transition metal ion found for (f) is
Explanation of Solution
Ground-state electronic configuration of the given ion (f) is,
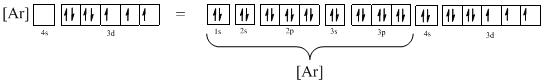
The atomic ground-state configuration is given in the problem statement. From this we can find that the given ion has an empty 4s orbital and 3d sub-shell that has seven electrons.
Identifying the ion as follows,
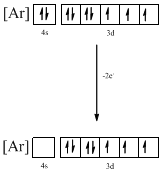
From the configuration given in the problem statement, we can conclude that the ion is formed by removing two electrons from 4s orbital. Therefore, there is only one possibility for the given ground-state electronic configuration. The ion is found to be
Want to see more full solutions like this?
Chapter 4 Solutions
EBK CHEMISTRY: ATOMS FIRST
- (a) Explain why the sizes of atoms change when proceeding across a period of the periodic table. (b) Explain why the sizes of transition metal atoms change very little across a period.arrow_forwardUse electron configurations to explain why (a) sulfur has a lower electron affinity than chlorine. (b) boron has a lower first ionization energy than beryllium. (c) chlorine has a lower first ionization energy than fluorine. (d) oxygen has a lower first ionization energy than nitrogen. (e) iodine has a lower electron affinity than bromine.arrow_forwardWrite electron configurations for the following elements. a. The Group III A element in the same period as 4Be b. The Period 3 element in the same group as 5B c. The lowest-atomic-numbered metal in Group IIA d. The two Period 3 elements that have no unpaired electronsarrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





