
Principles of General, Organic, Biological Chemistry
2nd Edition
ISBN: 9780073511191
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 4, Problem 4.31UKC
Consider the cooling curve drawn below.
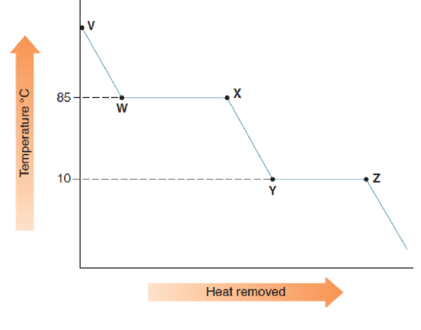
- a. Which line segment corresponds to the following changes of state?
- b. What is the melting point of the substance?
- c. What is the boiling point of the substance?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Principles of General, Organic, Biological Chemistry
Ch. 4.1 - Prob. 4.1PCh. 4.1 - Prob. 4.2PCh. 4.1 - Prob. 4.3PCh. 4.1 - Prob. 4.4PCh. 4.2 - Prob. 4.5PCh. 4.2 - Prob. 4.6PCh. 4.3 - Prob. 4.7PCh. 4.3 - Prob. 4.8PCh. 4.3 - Prob. 4.9PCh. 4.3 - Prob. 4.10P
Ch. 4.3 - Which of the compounds in each pair has stronger...Ch. 4.4 - Prob. 4.12PCh. 4.4 - Prob. 4.13PCh. 4.5 - A student has two containers one with 10 g of...Ch. 4.5 - Prob. 4.15PCh. 4.5 - How much energy is required to heat 28.0 g of iron...Ch. 4.5 - Prob. 4.17PCh. 4.5 - Prob. 4.18PCh. 4.5 - Prob. 4.19PCh. 4.6 - Prob. 4.20PCh. 4.6 - Prob. 4.21PCh. 4.6 - Label each process as endothermic or exothermic...Ch. 4.6 - Prob. 4.23PCh. 4.7 - Answer the following questions about the graph. a....Ch. 4.7 - Prob. 4.25PCh. 4.7 - If the cooling curve in Figure 4.5 represented a...Ch. 4.7 - How much energy (in calories) is released when...Ch. 4.7 - Prob. 4.28PCh. 4 - What phase change is shown in the accompanying...Ch. 4 - What phase change is shown in the accompanying...Ch. 4 - Consider the cooling curve drawn below. a. Which...Ch. 4 - Which line segments on the cooling curve in...Ch. 4 - Prob. 4.33UKCCh. 4 - Prob. 4.34UKCCh. 4 - Prob. 4.35UKCCh. 4 - Prob. 4.36UKCCh. 4 - Prob. 4.37UKCCh. 4 - Prob. 4.38UKCCh. 4 - Prob. 4.39APCh. 4 - Prob. 4.40APCh. 4 - Prob. 4.41APCh. 4 - Prob. 4.49APCh. 4 - Prob. 4.50APCh. 4 - Prob. 4.54APCh. 4 - Prob. 4.55APCh. 4 - Prob. 4.56APCh. 4 - Prob. 4.57APCh. 4 - Prob. 4.58APCh. 4 - Prob. 4.59APCh. 4 - Prob. 4.60APCh. 4 - Prob. 4.61APCh. 4 - Prob. 4.62APCh. 4 - Prob. 4.63APCh. 4 - Prob. 4.64APCh. 4 - Prob. 4.65APCh. 4 - Prob. 4.66APCh. 4 - Prob. 4.67APCh. 4 - Prob. 4.68APCh. 4 - Prob. 4.69APCh. 4 - Prob. 4.70APCh. 4 - Prob. 4.71APCh. 4 - Prob. 4.72APCh. 4 - Prob. 4.73APCh. 4 - Prob. 4.74APCh. 4 - Prob. 4.75APCh. 4 - Prob. 4.76APCh. 4 - Prob. 4.77APCh. 4 - Prob. 4.78APCh. 4 - Prob. 4.79APCh. 4 - Why does steam form when hot lava falls into the...Ch. 4 - Prob. 4.81APCh. 4 - Prob. 4.82AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Heat is added to boiling water. Explain why the temperature of the boiling water does not change. What does change?arrow_forwardWhat is the relationship between the intermolecular forces in a solid and its melting temperature?arrow_forwardWhat types of liquids typically form amorphous solids?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Viscosity, Cohesive and Adhesive Forces, Surface Tension, and Capillary Action; Author: Professor Dave Explains;https://www.youtube.com/watch?v=P_jQ1B9UwpU;License: Standard YouTube License, CC-BY