
Biology: Life on Earth with Physiology (11th Edition)
11th Edition
ISBN: 9780133923001
Author: Gerald Audesirk, Teresa Audesirk, Bruce E. Byers
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3.6, Problem 1CT
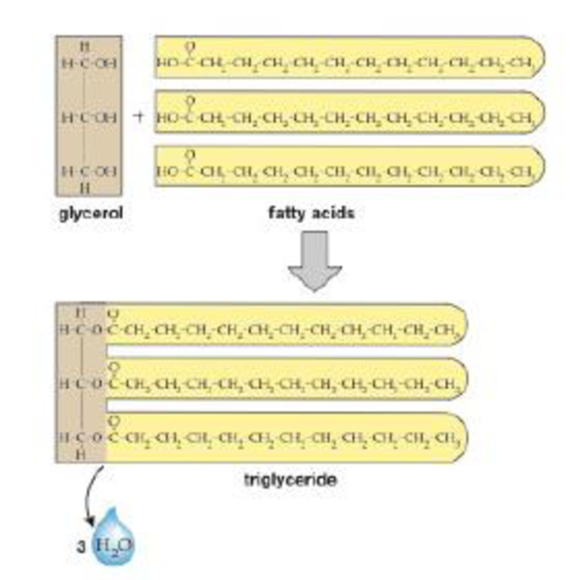
FIGURE 3-22 Synthesis of a triglyceride Dehydration synthesis links a single glycerol molecule with three fatty acids to form a triglyceride and three water molecules.
THINK CRITICALLY what Kind of reaction tweaks this molecule apart?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What are biofertilizers and mention the significance
PCBs and River Otters: Otters in Washington State’s Green-Duwamish River have high levels of polychlorinated biphenyls (PCBs) in their livers. PCBs can bind to the estrogen receptors in animals and disrupt the endocrine system of these otters. The PCBs seem to increase the estrogen to androgen ratio, skewing the ratio toward too much estrogen.
How would increased estrogen affect the river otter population?
Based on your reading of the materials in this unit, what factors can affect fertility in humans?
Explain how each of the factors affecting human fertility that you described can disrupt the human endocrine system to affect reproduction.
Other than oil and alcohol, are there other liquids you could compare to water (that are liquid at room temperature)?
How is water unique compared to these other liquids?
What follow-up experiment would you like to do, and how would you relate it to your life?
Chapter 3 Solutions
Biology: Life on Earth with Physiology (11th Edition)
Ch. 3.1 - define organic molecules and explain why carbon is...Ch. 3.1 - which of these is/are polar molecules? (you may...Ch. 3.1 - explain why functional groups are important in...Ch. 3.1 - name and describe the properties of seven...Ch. 3.2 - define organic molecules and explain why carbon is...Ch. 3.3 - describe the major types of carbohydrates?Ch. 3.3 - A 19-year old 6' 2' male weighing 297 pounds comes...Ch. 3.3 - Describe hydrolysis of this molecule.Ch. 3.3 - provide examples of each type of carbohydrate and...Ch. 3.4 - Infectious prions such as those that cause mad cow...
Ch. 3.4 - describe protein subunits and how proteins are...Ch. 3.4 - Look up the rest of the amino acids and. based on...Ch. 3.4 - explain the four levels of protein structure and...Ch. 3.4 - Why do many proteins, when heated excessively....Ch. 3.4 - list several functions of proteins and provide...Ch. 3.4 - describe the properties of Intrinsically...Ch. 3.5 - Puzzling Proteins All cells use DNA as a blueprint...Ch. 3.5 - describe the general structure of nucleotides?Ch. 3.5 - Why a Perm Is (Temporarily) Permanent?Ch. 3.5 - list three different functions of nucleotides?Ch. 3.5 - explain how nucleic acids are synthesized?Ch. 3.5 - give two examples of nucleic acids and their...Ch. 3.6 - Prob. 1CSRCh. 3.6 - FIGURE 3-22 Synthesis of a triglyceride...Ch. 3.6 - compare and contrast the structure and synthesis...Ch. 3.6 - An obese 55-year-old woman consults her physician...Ch. 3.6 - What kind of reaction breaks this molecule apart?Ch. 3.6 - why are steroid hormones able to diffuse through...Ch. 3.6 - describe the functions of fats, oils, and waxes?Ch. 3.6 - Why are steroid hormones able to diffuse through...Ch. 3.6 - Prob. 3CYLCh. 3 - Based on their structure, sketch and explain how...Ch. 3 - In organic molecules made of chains of subunits,...Ch. 3 - Polar molecules a. dissolve in lipids. b. are...Ch. 3 - Prob. 1RQCh. 3 - Prob. 2ACCh. 3 - Prob. 2FIBCh. 3 - Prob. 2MCCh. 3 - List the four principal classes of biological...Ch. 3 - Prob. 3ACCh. 3 - Prob. 3FIBCh. 3 - Prob. 3MCCh. 3 - Prob. 3RQCh. 3 - Prob. 4FIBCh. 3 - Which of the following is not composed of...Ch. 3 - Prob. 4RQCh. 3 - Fill in the following with the appropriate type of...Ch. 3 - Prob. 5MCCh. 3 - Prob. 5RQCh. 3 - Describe the synthesis of a protein from amino...Ch. 3 - Where in nature do we find cellulose? Where do we...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Selection of Traits What adaptations do scavengers have for locating and feeding on prey? What adaptations do predators have for capturing and consuming prey?arrow_forwardCompetition Between Species What natural processes limit populations from growing too large? What are some resources organisms can compete over in their natural habitat?arrow_forwardSpecies Interactions Explain how predators, prey and scavengers interact. Explain whether predators and scavengers are necessary or beneficial for an ecosystem.arrow_forward
- magine that you are conducting research on fruit type and seed dispersal. You submitted a paper to a peer-reviewed journal that addresses the factors that impact fruit type and seed dispersal mechanisms in plants of Central America. The editor of the journal communicates that your paper may be published if you make ‘minor revisions’ to the document. Describe two characteristics that you would expect in seeds that are dispersed by the wind. Contrast this with what you would expect for seeds that are gathered, buried or eaten by animals, and explain why they are different. (Editor’s note: Providing this information in your discussion will help readers to consider the significance of the research).arrow_forwardWhat is the difference between Uniporters, Symporters and Antiporters? Which of these are examples of active transport?arrow_forwardWhat are coupled transporters?arrow_forward
- How do histamine and prostaglandins help in the mobilization of leukocytes to an injury site? What are chemotactic factors? How do they affect inflammation process?arrow_forwardCompare and contrast neutrophils and macrophages. Describe two ways they are different and two ways they are similar.arrow_forwardDescribe the effects of three cytokines (not involved in the initial inflammation response). What cells release them?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning

Biology Today and Tomorrow without Physiology (Mi...
Biology
ISBN:9781305117396
Author:Cecie Starr, Christine Evers, Lisa Starr
Publisher:Cengage Learning

Biology (MindTap Course List)
Biology
ISBN:9781337392938
Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:Cengage Learning
Macromolecules | Classes and Functions; Author: 2 Minute Classroom;https://www.youtube.com/watch?v=V5hhrDFo8Vk;License: Standard youtube license