
Concept explainers
(a)
Interpretation:
The name, condensed structural formula and molecular mass of the given molecule should be determined.
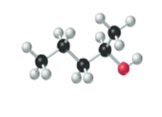
Concept introduction:
Name of the compound can be assigned according to below rule:
First, identify the number of carbon atoms present in chain, and then identify the
Condensed structural formula is defined as the structural formula which shows the spatial arrangement of bonds in an organic compound. In this structural formula, the bonds between the atoms are shown with lines and omit the bond between carbon and hydrogen atoms.
Molecular mass is defined as the sum of mass of each atom present in the compound.
(b)
Interpretation:
The name, condensed structural formula and molecular mass of the given molecule should be drawn.
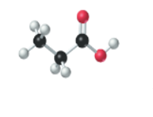
Concept introduction:
Name of the compound can be assigned according to below rule:
First, identify the number of carbon atoms present in chain, and then identify the function group if present in the given compound.
Condensed structural formula is defined as the structural formula which shows the spatial arrangement of bonds in an organic compound. In this structural formula, the bonds between the atoms are shown with lines and omit the bond between carbon and hydrogen atoms.
Molecular mass is defined as the sum of mass of each atom present in the compound.
(c)
Interpretation:
The name, condensed structural formula and molecular mass of the given molecule should be drawn.
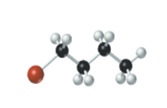
Concept introduction:
Name of the compound can be assigned according to below rule:
First, identify the number of carbon atoms present in chain, and then identify the function group if present in the given compound.
Condensed structural formula is defined as the structural formula which shows the spatial arrangement of bonds in an organic compound. In this structural formula, the bonds between the atoms are shown with lines and omit the bond between carbon and hydrogen atoms.
Molecular mass is defined as the sum of mass of each atom present in the compound.
(d)
Interpretation:
The name, condensed structural formula and molecular mass of the given molecule should be drawn.
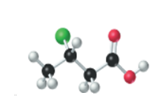
Concept introduction:
Name of the compound can be assigned according to below rule:
First, identify the number of carbon atoms present in chain, and then identify the function group if present in the given compound.
Condensed structural formula is defined as the structural formula which shows the spatial arrangement of bonds in an organic compound. In this structural formula, the bonds between the atoms are shown with lines and omit the bond between carbon and hydrogen atoms.
Molecular mass is defined as the sum of mass of each atom present in the compound.
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
GENERAL CHEMISTRY-MOD.MASTERINGCHEM.
- Assume that the ratio of moles of Cl to moles of Cu are 2.7. If we can represent the formula by CuClx, what is the value for x? Round your answer to the nearest whole number. Do NOT include units. So, if you think the formula is CuCl6, you enter the number 6.arrow_forwardCalculate the percent oxygen by mass in disilicon trioxide(Si2O3) to 3 significant figures.arrow_forwardComplete the table below for calculating the molar mass of the ionic compound silver sulfide. Formula Cation Ag+ Anion S²- Molar mass of ion g/mol g/mol Number of ions mol mol Molar mass silver sulfide = = = Mass of ion in one mole of silver sulfide g/mol g garrow_forward
- An organic compound was analyzed by mass spectrometry and found to contain 19.488% carbon, 2.453% hydrogen, and the rest sulfur. What is the compound's empirical formula? Enter a number in each space. Answers must be numerical - for example, enter "1" not "one".arrow_forwardFinding mass percent from chemical formulae This is the chemical formula for diethyl ether: (CH,CH,),O. О. Calculate the mass percent of carbon in diethyl ether. Round your answer to the nearest percentage. 0%arrow_forwardA compound has a molar mass of 28.06 g. What is its chemical formula if carbon and hydrogen are present in the compound in a 1-to-2 molar ratio? Use underscore to indicate subscripts. For example: H2O would be, H_2Oarrow_forward
- 3- calculate the number of ( 0) atom in 1.5 g of glucose (C;H1206).arrow_forwardBased on the following structural formulas, calculate thepercentage of carbon by mass present in each compound:arrow_forwardA 3.260 gram sample of an organic compound containing C, H and O is analyzed by combustion analysis and 5.809 grams of CO2 and 2.379 grams of H20 are produced. In a separate experiment, the molar mass is found to be 74.08 g/mol. Determine the empirical formula and the molecular formula of the organic compound. Enter the elements in the order C, H, O empirical formula = molecular formula = Give the balanced equation for each of the following.arrow_forward
- When 1.615 grams of a hydrocarbon, CxHy, were burned in a combustion analysis apparatus, 5.545 grams of CO2 and 0.9082 grams of H₂O were produced. In a separate experiment, the molar mass of the compound was found to be 128.2 g/mol. Determine the empirical formula and the molecular formula of the hydrocarbon. Enter the elements in the order presented in the question. empirical formula = molecular formula = =arrow_forwardMass Spectrometry of Molecular Compounds Mass spectrometry is not only used for isotopic evaluation of the elements. It can also be used for identification of molecular samples as well. Consider, for example, the mass spectrum of pentane (Figure 5): Lifie relative abundance 57 29 72 10 20 30 40 50 60 70 miz Figure 5. Pentane Mass Spectrum The pentane molecule (CH;CH,CH;CH;CH; or C;H12) is a straight-chain hydrocarbon. In Figure 5, notice there are four groups of peaks. This forms what is known as the cracking pattern of the molecule. The first peak is at m/z = 72, corresponding to the molecular ion, C;H,2", where only one electron was knocked off the pentane molecule. The three peak groups are from fragments of pentane created in the ionizer: m/z = 57 (CH;CH,CH,CH,"), m/z = 43 (CH;CH,CH;"), m/z = 29 (CH;CH,"). These species have had pieces of the molecule knocked off in addition to losing an electron. 4. For the peak at m/z = 43, what part of the pentane molecule was knocked off? Use…arrow_forwardyou are to determine the molecular formula for a molecular compound consisting of only carbon & hydrogen. The molar mass of this compound is 42.08 g/mol. Part D: The empirical formula has a molar mass of 14.026 g/mol (this is called the empirical molar mass). Divide the molar mass for the total compound given above by the empirical formula molar mass. This will give you a number for multiplying the carbon atoms and hydrogen atoms in the empirical formula. What is the multiplication value?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
