
Organic Chemistry - Standalone book
10th Edition
ISBN: 9780073511214
Author: Francis A Carey Dr., Robert M. Giuliano
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 42P
One of the following two stereoisomers is
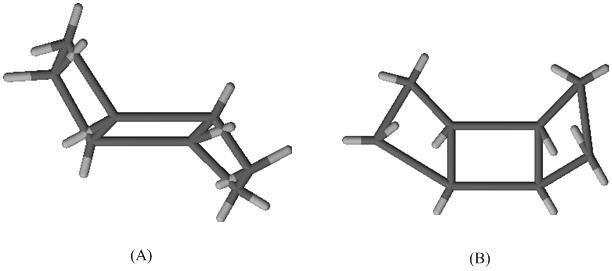
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Identify the relationship between the given pairs of compounds. Choose from:
A = enantiomers
diastereomers
C = constitutional isomers
D = geometric isomers
E = conformational isomers
F = two molecules of the same compound
Br
Br
Br
Br
The barrier of rotation for the C–C bond in bromoethane is 3.7 kcal/mol. We previously learned that an eclipsing C–H bond with another C–H is about 1 kcal/mol each. Based on this, what is the energy cost to eclipse a C–H bond with a C–Br bond?
The torsional energy in propane is 14 kJ/mol (3.4 kcal/mol). Because each H,H eclipsing interaction is worth 4.0 kJ/mol (1.0 kcal/mol) of destabilization, how much is one H,CH3 eclipsing interaction worth in destabilization?
Chapter 3 Solutions
Organic Chemistry - Standalone book
Ch. 3.1 - Identify the alkanes corresponding to each of the...Ch. 3.1 - Find the conformations in Figure 3.4 in which the...Ch. 3.2 - Sketch a potential energy diagram for rotation...Ch. 3.2 - Acetylcholine is a neurotransmitter in the central...Ch. 3.2 - Prob. 5PCh. 3.5 - The heats of combustion of ethylcyclopropane and...Ch. 3.8 - Prob. 7PCh. 3.10 - The following questions relate to a cyclohexane...Ch. 3.10 - Draw the most stable conformation of...Ch. 3.11 - Prob. 10P
Ch. 3.11 - Prob. 11PCh. 3.12 - Based on what you know about disubstituted...Ch. 3.12 - Write structural formulas for the most stable...Ch. 3.14 - Cubane (C4H8) is the common name of the polycyclic...Ch. 3.14 - Prob. 15PCh. 3.14 - Prob. 16PCh. 3.14 - Prob. 17PCh. 3.14 - Prob. 18PCh. 3.15 - Prob. 19PCh. 3 - Give the IUPAC names of each of the following: (a)...Ch. 3 - Draw Newman projections for the gauche and...Ch. 3 - Identify all atoms that are (a) anti and (b)...Ch. 3 - Prob. 23PCh. 3 - Prob. 24PCh. 3 - Prob. 25PCh. 3 - Prob. 26PCh. 3 - Prob. 27PCh. 3 - Prob. 28PCh. 3 - Oxidation of 4-tert-butylthiane proceeds according...Ch. 3 - The following are representations of two forms of...Ch. 3 - Draw (a) a Newman projection of the most stable...Ch. 3 - Write a structural formula for the most stable...Ch. 3 - Sight down the C-2-C-3 bond, and draw Newman...Ch. 3 - Prob. 34PCh. 3 - Sketch an approximate potential energy diagram for...Ch. 3 - Prob. 36PCh. 3 - Even though the methyl group occupies an...Ch. 3 - Which do you expect to be the more stable...Ch. 3 - Arrange the trimethylcyclohexane isomers shown in...Ch. 3 - Identify the more stable stereoisomer in each of...Ch. 3 - One stereoisomer of 1,1,3,5-tetramethylcyclohexane...Ch. 3 - One of the following two stereoisomers is...Ch. 3 - In each of the following groups of compounds,...Ch. 3 - The heats of combustion of the more and less...Ch. 3 - The measured dipole moment of ClCH2CH2Cl is 1.12D....Ch. 3 - Prob. 46PCh. 3 - Prob. 47PCh. 3 - Prob. 48DSPCh. 3 - Prob. 49DSPCh. 3 - Prob. 50DSPCh. 3 - Prob. 51DSPCh. 3 - Prob. 52DSPCh. 3 - Prob. 53DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Label each pair of compounds as constitutional isomers, stereoisomers, or not isomers of each other.arrow_forwardClassify the pair of compounds as the same compound, enantiomers, diastereomers, constitutional isomers, or not isomeric. Also, select the correct IUPAC name, including the correct (R) or (S) designation, for each. H Br Br CH3 H H3C Br H Br Br Br Compound 1 Compound 2 The compounds are identical diastereomers not isomeric constitutional isomers enantiomers The correct IUPAC names are: Compound 1: (2R,3R)-1,2,3-tribromobutane, Compound 2: (2R,3R)-1,2,3-tribromobutane Compound 1: (2R,3S)-1,2,3-tribromobutane, Compound 2: (2S,3S)-1,2,3-tribromobutane Compound 1: (2S,35)-1,2,3-tribromobutane, Compound 2: (2R,3S)-1,2,3-tribromobutane Compound 1: (2R,3R)-1,2,3-tribromobutane, Compound 2: (2S,3R)-1,2,3-tribromobutanearrow_forwardStereoisomers share the same connectivity and differ only in the way their atoms are arranged in space. Draw the structure of a compound that is a stereoisomer ofarrow_forward
- What is the relationship between the two? - enantiomers - conformers - constitutional isomers - conformers - identical - resonance formsarrow_forwardWhat is the chiral center in each compound. R or Sarrow_forwardIndicate whether the pair of structures shown represent stereoisomers, constitutional isomers, different conformations of the same compound, or the same conformation of a compound viewed from a different perspective. Note that cis, trans isomers are an example of stereoisomers. Br Br Br 4 Br -Br Br Br 22arrow_forward
- Indicate whether the pair of structures shown represent stereoisomers, constitutional isomers, different conformations of the same compound, or the same conformation of a compound viewed from a different perspective. Note that cis, trans isomers are an example of stereoisomers. H3C. Br CI HO. CH3 H3C. Br CH3 Br CH3 Br CH3 CI H3C- CH3 HO- CH3 CI -CH(CH3)2 Br CH3 CH3 -Br CH3 CI CH(CH3)2arrow_forwardClassify the following as constitutional isomers, enantiomers, the same compound, or not isomers. CH3 H₂N V H3C 4 The two compounds are: not isomers constitutional isomers same compound CH3 enantiomers CH3 H3C H₂ H H3Carrow_forward1a. How many stereogenic centers are present 1c. Draw a three-dimensional structure of a in the structure below? Indicate them with asterisk(s). How many stereoisomers stereoisomers are possible? chiral compound with the molecular formula of C4H4Cl₂ that does not have a stereogenic carbon. In addition, draw the enantiomer of this compound. Number of stereogenic centers: Number of stereoisomers possible: 1b. Draw one of the two most stable stereoisomers of the compound in 1a using a planar structure with wedges and dashes. Now draw it in its preferred chair conformation. 1d. Draw two meso compounds with the molecular formula of C7H14.arrow_forward
- 7. Draw the two chair conformations for the molecule below. Given the corresponding energy values, place the conformations in the appropriate boxes. Substituent AG° (eq→ ax) (kcal/mol) -CN -CH, 0.2 NEC 1.7 less stable chair more stable chair Clearly explain why the energy barrier associated with a nitrile group (-CN) moving from an equatorial position to an axial position is much lower than for a methyl group (-CH3).arrow_forwardTwo disubstituted cyclohexane molecules are depicted. Classify the pair as the same compound, enantiomers, diastereomers, constitutional isomers, or not isomeric. CH3 CH3 and H3C ČH3 The compounds are: the same compound not isomeric enantiomers constitutional isomers diastereomersarrow_forwardConsider the following pairs of structures. Identify the relationship between them by describing them as representing enantiomers, diastereomers, constitutional isomers, or two molecules of the same compound. H H3C" CH3 Br Hitm Br enantiomers constitutional isomers diastereomers H B H F F two molecules of the same compound CH3 H Ba CH3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License