
Organic Chemistry - Standalone book
10th Edition
ISBN: 9780073511214
Author: Francis A Carey Dr., Robert M. Giuliano
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Question
Chapter 3, Problem 26P
Interpretation Introduction
Interpretation:
In each case, it is to be specified whether the designated substituent is axial or equatorial for the given steroid skeleton having a cis ring fusion for the first two rings.
Concept introduction:
The position of axial and equatorial bonds in cyclohexane chair form is
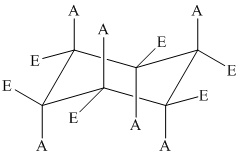
If the two substituents are on the same side of the ring, then they are cis to each other.
If the two substituents are on the opposite side of the ring, then they are trans to each other.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
How many asymmetric carbons and stereoisomers are there for an aldohexose? For aketohexose?
What are the configurations of all chiral centers of the attached molecule?
Please state the answer in clear words along with any pictures/reactions.
Please also point out where the chiral centers are.
Chapter 3 Solutions
Organic Chemistry - Standalone book
Ch. 3.1 - Identify the alkanes corresponding to each of the...Ch. 3.1 - Find the conformations in Figure 3.4 in which the...Ch. 3.2 - Sketch a potential energy diagram for rotation...Ch. 3.2 - Acetylcholine is a neurotransmitter in the central...Ch. 3.2 - Prob. 5PCh. 3.5 - The heats of combustion of ethylcyclopropane and...Ch. 3.8 - Prob. 7PCh. 3.10 - The following questions relate to a cyclohexane...Ch. 3.10 - Draw the most stable conformation of...Ch. 3.11 - Prob. 10P
Ch. 3.11 - Prob. 11PCh. 3.12 - Based on what you know about disubstituted...Ch. 3.12 - Write structural formulas for the most stable...Ch. 3.14 - Cubane (C4H8) is the common name of the polycyclic...Ch. 3.14 - Prob. 15PCh. 3.14 - Prob. 16PCh. 3.14 - Prob. 17PCh. 3.14 - Prob. 18PCh. 3.15 - Prob. 19PCh. 3 - Give the IUPAC names of each of the following: (a)...Ch. 3 - Draw Newman projections for the gauche and...Ch. 3 - Identify all atoms that are (a) anti and (b)...Ch. 3 - Prob. 23PCh. 3 - Prob. 24PCh. 3 - Prob. 25PCh. 3 - Prob. 26PCh. 3 - Prob. 27PCh. 3 - Prob. 28PCh. 3 - Oxidation of 4-tert-butylthiane proceeds according...Ch. 3 - The following are representations of two forms of...Ch. 3 - Draw (a) a Newman projection of the most stable...Ch. 3 - Write a structural formula for the most stable...Ch. 3 - Sight down the C-2-C-3 bond, and draw Newman...Ch. 3 - Prob. 34PCh. 3 - Sketch an approximate potential energy diagram for...Ch. 3 - Prob. 36PCh. 3 - Even though the methyl group occupies an...Ch. 3 - Which do you expect to be the more stable...Ch. 3 - Arrange the trimethylcyclohexane isomers shown in...Ch. 3 - Identify the more stable stereoisomer in each of...Ch. 3 - One stereoisomer of 1,1,3,5-tetramethylcyclohexane...Ch. 3 - One of the following two stereoisomers is...Ch. 3 - In each of the following groups of compounds,...Ch. 3 - The heats of combustion of the more and less...Ch. 3 - The measured dipole moment of ClCH2CH2Cl is 1.12D....Ch. 3 - Prob. 46PCh. 3 - Prob. 47PCh. 3 - Prob. 48DSPCh. 3 - Prob. 49DSPCh. 3 - Prob. 50DSPCh. 3 - Prob. 51DSPCh. 3 - Prob. 52DSPCh. 3 - Prob. 53DSP
Knowledge Booster
Similar questions
- Trehalose and isomaltose are both dimers of glucose. However, they have considerablydifferent reactivities. Concisely explain why these differences are observed.arrow_forwardWhich of the attached cyclic molecules are meso compounds?arrow_forwardFor the following structure, draw and indicate the low and high-energy chair conformations and calculate the energy difference between the 2 conformations using the table provided.arrow_forward
- To the following statement, answer true or false and explain your answer. Q)To be meso, a molecule must have at least two chiral centersarrow_forwardWhat is the maximum number of stereoisomers possible for the attached compound?arrow_forwardWhat are all the steroisomers of this compound?arrow_forward
- Use the following wedge formula mentioned in attached diagram to answer the following question. Draw one enantiomer and two diastereomers of structure (ii).arrow_forwardWhich molecule would have their 2 chair conformations be of equal energies?arrow_forwardBest describes the relationship between the straight chain forms of D-glucose and D-galactose, given that they are non-cyclic molecules that differ only in stereochemical configuration at the fourth carbon?arrow_forward
- Mark all chiral carbons with a star in the following molecules.arrow_forwarda) For each of the following pairs of compounds, identify them as identical, enantiomers or Note that there are three pairs of compounds. b) Define the stereochemistry (R or S) at all the chiral carbons for only one member of each pair of compounds.arrow_forwardIndicate, with asterisks (*), all of the chiral centers in each of the following molecules. For eachmolecule, what is the maximum number of stereoisomers that can be formed?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning