
Concept explainers
(a)
Plot of wave function as a function of
(a)
Answer to Problem 74P
The wave function was plotted as a function of
Explanation of Solution
Write the given wave function of the electron.
Here,
Write the formula to calculate the probability of finding a particle in a certain range.
Here,
Refer equation (I) and plot the wave function as a function of
Figure 1 below shows the plot of wave function.
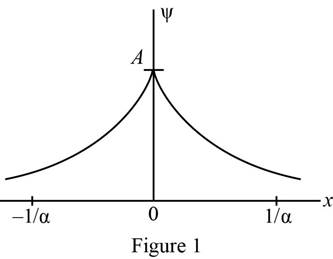
Conclusion:
The wave function was plotted as a function of
(b)
Plot of probability density as a function of
(b)
Answer to Problem 74P
The probability density was plotted as a function of
Explanation of Solution
Write the given wave function of the electron.
Here,
Write the formula to calculate the probability of finding a particle in a certain range.
Here,
Refer equation (II) and plot the probability density as a function of
Figure 2 below shows the plot of wave function.
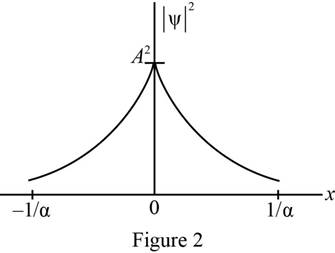
Conclusion:
The probability density was plotted as a function of
(c)
To show that
(c)
Answer to Problem 74P
The
Explanation of Solution
Write the given wave function of the electron.
Here,
For the wave function to be a reasonable wave function, there are set of condition.
The
As
The
The
Conclusion:
The
(d)
To normalize the wave function.
(d)
Answer to Problem 74P
The normalization constant of the given wave function is
Explanation of Solution
Write the given wave function of the electron.
Here,
Write the condition for normalized wave function.
The wave function is symmetric. Thus re-write the above condition.
Substitute equation (I) in the above equation.
Conclusion:
The normalization constant of the given wave function is
(e)
The probability of finding the electron in the range
(e)
Answer to Problem 74P
The probability of finding the particle in the range
Explanation of Solution
Refer section (d) and write the given normalized wave function of the electron.
Here,
Write the formula to calculate the probability of finding a particle in a certain range.
Here,
Refer equation (II) in equation (III) to determine probability in range
Conclusion:
The probability of finding the particle in the range
Want to see more full solutions like this?
Chapter 28 Solutions
Principles of Physics: A Calculus-Based Text
- If a classical harmonic oscillator can at rest, why can the quantum harmonic oscillator never be at rest? Does this violate Bohr 's correspondence principle?arrow_forwardCan the de Broglie wavelength of a particle be known precisely? Can the position of a particle be known precisely?arrow_forwardThrough what minimum potential difference must an electron in an x-ray tube be accelerated so that it can produce x rays with a wavelength of 0.100 nm?arrow_forward
- Suppose the minimum uncertainty in the position of a particle is equal to its de Broglie wavelength. If the particle has an average speed of 4.5 x 10^5 m/s, what is the minimum uncertainty in its speed?arrow_forwardHow much the angular momentum of this electron becomes if it absorbed an electromagnetic wave with frequency (8.21 x 104 Hz)?arrow_forwardA Quantum system has a ground state with energy E0 = 100meV and a 3-fold degenerate excited state with energy E1 = 100meV . Calculate the probability of finding the system in its groud state when it is at T = 300 K? a) 0.94 b) 0.06 c) 1 d) 0.98arrow_forward
- Calculate the longest wavelength of light that can eject electrons from a surface with a work function of 2.46 eV.arrow_forwardI have a physics question as follows: An isolated copper sphere of radius 5.55 cm, initially uncharged, is illuminated by ultraviolet light of wavelength 247 nm. The work function for copper is 4.70 eV. What charge does the photoelectric effect induce on the sphere?arrow_forwardAn electron is trapped in a rectangular region of length x = 1.25 nm and width y = 2.76 nm. What isthe minimum value that might be measured for its kinetic energy?arrow_forward
- What wavelengths are possible when an electron comes from the second excited state to the ground state such as an H atom? and what would be the momenta of the photons?arrow_forwardThe ground-state energy of a harmonic oscillator is 5.60 eV. If the oscillator undergoes a transition from its n = 3 to n = 2 level by emitting a photon, what is the wavelength of the photon?arrow_forwardShow that the wave function ψ = Ae i(i-ωt) is a solution to the Schrödinger equation (as shown), where k = 2π/λ and U = 0.arrow_forward
 University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax
University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning
An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning




