
Concept explainers
(a)
Interpretation:
One alternative tautomeric structure for Adenine has to be written.
Concept Introduction:
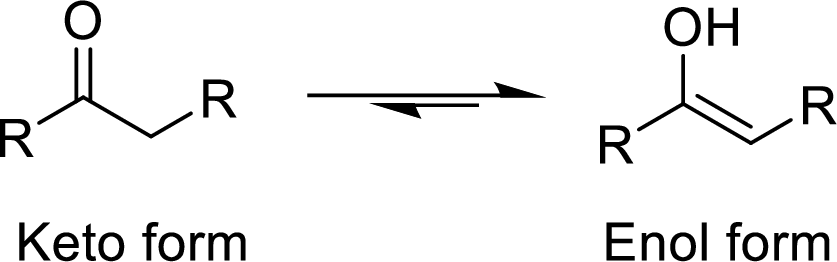
The isomers which differ in the position of the protons and electrons and the carbon skeleton is same are said to be Tautomers and the mechanism is known as Tautomerism. The common example for Tautomerism is keto-enol tautomerization. The keto group is converted to enol (alcohol) group by the intramolecular exchange of protons.
(b)
Interpretation:
Will the tautomeric structure of Adenine base pairs with Thymine or any other base has to be given.
Concept Introduction:
In the DNA double helix, the bases are paired with each other on the adjacent strands to hold the double helix. The bases present in DNA are Adenine, Guanine, Cytosine and Thymine. The pairing occurs between the Adenine and Thymine (A-T) and Guanine and Cytosine (G-C).
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry
- On complete hydrolysis, a polypeptide gives two alanine, one leucine, one methionine, one phenylalanine, and one valine residue. Partial hydrolysis gives the following fragments: Ala-Phe, Leu-Met, Val-Ala, Phe-Leu. It is known that the first amino acid in the sequence is valine and the last one is methionine. What is the complete sequence of amino acids?arrow_forwardDo the amino acid sequences: valine-asparagine and asparagine-valine represent the same compound? Explain.arrow_forwardAlthough RNA is single-stranded, the strand sometimes folds back on itself to give a complementary portion. What would be the complementary portion of the RNA fragment having the bases uracil-cytosine-guanine? A virus is a particle that contains DNA or RNA that is surrounded by a coat of protein that can replicate only in a host cell. The influencza virus shown in this electron microscope image is an RNA virus.arrow_forward
- The sequence of a short DNA segment is ATGGCAATAC. (a) What name do we give to the two ends (terminals) of a DNA molecule? (b) In this segment, which end is which? (c) What would be the sequence of the complementary strand?arrow_forwardErwin Chargaff’s discovery that DNA contains equimolar amounts of guanine and cytosine and also equimolar amounts ofadenine and thymine has come to be known as Chargaff’s rule:G = C and A = T(a) Does Chargaff’s rule imply that equal amounts of guanine and adenine are present in DNA? That is, does G = A?(b) Does Chargaff’s rule imply that the sum of the purine residues equals the sum of the pyrimidine residues? That is,does A + G = C + T?(c) Does Chargaff’s rule apply only to double-stranded DNA, or would it also apply to each individual strand if thedouble helical strand were separated into its two complementary strands?arrow_forwardAs we’ve discussed, a peptide bond is made when amino group of one amino acid combines with the carboxylic acid group of another amino acid (releasing a water molecule in the process). The C-N bond formed in this process is called a peptide bond. Peptide bonds have a few properties that might be unexpected. b) Another observation is that although the N-H of the peptide bond is able to serve as a H-bond donor the N atom of the peptide bond does not serve as an effective H-bond acceptor. Provide an explanation for this observation, using Lewis structures, VSEPR theory and/or valence bond theory as appropriate.c) It is also fairly accepted that while proteins undergo dynamic motions and conformational changes, and while R groups can freely rotate about Cα, it is generally not possible to freely rotate around a peptide bond. Please explain this observation, using a picture showing relevant orbitals on relevant atoms (your answer for part (a) might provide a useful basis for your reasoning…arrow_forward
- C) NUCLEIC ACID Sketch the general structure of a nucleic acid monomer and name all characteristic chemical groups: TETAAL Sketch the general structure of a nucleic acid polymer and name all characteristic chemical group: Name of bond between monomers: D) LIPID Sketch the general structure of a triglyceride molecule, name the characteristic chemical groups that form the molecule, and label the chemical linkages between the groups: inte imveneg Sketch the general structure of a phospholipid molecule, name the characteristic chemical groups that form the mol- ecule, and show how phospholipids align to create cell membranes. Name the characteristic that distinguishes all lipids from other large molecules:arrow_forwardSelect the single best answer. Watson and Crick determined that the DNA molecule has two helical strands. Each strand is made up which consist of a base, a deoxyribose, and a phosphate group linked together. of What word most correctly fills in the blank in the preceding statement? nucleotides fatty acids monosaccharides amino acidsarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





