
Anatomy & Physiology (6th Edition)
6th Edition
ISBN: 9780134156415
Author: Elaine N. Marieb, Katja N. Hoehn
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 25, Problem 12MC
The pH of blood varies directly with (a) HCO3–, (b) 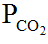 , (c) H+, (d) none of the above.
, (c) H+, (d) none of the above.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The pH of blood varies directly with (a) HCO3−, (b) PCO2, (c) H+, (d) none of the above.
An individual's ratio of carbonic acid and bicarbonate ions is 17/0.6
Which of the following is a true statement?
For a total of 3 answers - choose one of each of the following pairs:
i) respiratory or metabolic
ii) acidosis or alkalosis
iii) uncompensated or partial compensation
Harry ate a late meal at a cheap diner on the way home from work.A couple of hours later, he vomited three times, and then he consumed several packages of an antacid (mostly NaHCO3) over thenext several hours. By the evening of the next day, he was feelingbetter. During this ordeal, his blood pH did not deviate significantly.Select the mechanism(s) that helped maintain Harry’s pH within anormal range.(1) increased respiratory rate(2) increased H+ secretion by the renal tubule(3) decreased respiratory rate(4) decreased H+ secretion by the renal tubulea. 1 b. 2 c. 1,2 d. 3 e. 3,4
Chapter 25 Solutions
Anatomy & Physiology (6th Edition)
Ch. 25.1 - Which do you have more of, extracellular or...Ch. 25.1 - What is the major cation in the ECF? In ICF? What...Ch. 25.1 - If you eat salty pretzels without drinking, what...Ch. 25.2 - Prob. 6CYUCh. 25.2 - ADH, by itself, cannot reduce an increase in...Ch. 25.2 - For each of the following, state whether it might...Ch. 25.3 - Nathan has Addisons disease (insufficient...Ch. 25.3 - Prob. 9CYUCh. 25.3 - Prob. 10CYUCh. 25.4 - Define acidemia and alkalemia.
Ch. 25.4 - What are the bodys three major chemical buffer...Ch. 25.4 - Joanne, a diabetic patient, is at the emergency...Ch. 25.5 - Reabsorption of HCO3 is always tied to the...Ch. 25.5 - Prob. 14CYUCh. 25.5 - Prob. 15CYUCh. 25.5 - Prob. 16CYUCh. 25.6 - Which two abnormalities in plasma are key features...Ch. 25.6 - How do the kidneys compensate for respiratory...Ch. 25 - Body water content is greatest in (a) infants, (b)...Ch. 25 - Potassium, magnesium, and phosphate ions are the...Ch. 25 - Sodium balance is regulated primarily by control...Ch. 25 - Prob. 4MCCh. 25 - Two main substances regulated by the influence of...Ch. 25 - Two substances regulated by parathyroid hormone.Ch. 25 - Two substances secreted into the proximal...Ch. 25 - Prob. 8MCCh. 25 - Prob. 9MCCh. 25 - Prob. 10MCCh. 25 - Prob. 11MCCh. 25 - The pH of blood varies directly with (a) HCO3, (b)...Ch. 25 - In an individual with metabolic acidosis, a clue...Ch. 25 - Name the body fluid compartments, noting their...Ch. 25 - Prob. 2SAQCh. 25 - Prob. 3SAQCh. 25 - Prob. 4SAQCh. 25 - Prob. 5SAQCh. 25 - Explain how the chemical buffer systems resist...Ch. 25 - Explain the relationship of the following to renal...Ch. 25 - Mr. Heyden, a somewhat stocky 72-year-old man, is...Ch. 25 - Mr. Heyden, a somewhat stocky 72-year-old man, is...Ch. 25 - Mr. Heyden, a somewhat stocky 72-year-old man, is...Ch. 25 - Mr. Heyden, a somewhat stocky 72-year-old man, is...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- In what range would the blood pH of a heavy smoker with advanced emphysema fall: (a) less than 7.35; (b) 7.35–7.45; (c) greater than 7.45? Explain your choice.arrow_forwardCarbon dioxide dissolved in body fluids has a pronounced effect on pH.(a) Does pH go up or down when carbon dioxide dissolves in these fluids? Does this change indicate higher or lower acidity?(b) What does a blood gas analysis measure?arrow_forwarda 86 years old women brought to emergency room with these readings :PH = 7.22 (normal :7.35-7.45) , PaCO2 = 9.6 mmHg ( normal: 35-45) ,HCO3- = 3.9 mmol/l ( normal : 22-26) .. she complains from : A) Metabolic acidosis. B)Respiratory acidosis. C)Metabolic alkalosis. D)Respiratory alkalosis.arrow_forward
- A) how to explain that a venous blood looks purplish blue and an arterial blood appears bright red to us? B) what other important for life porphyrins (complex ions) do you know?arrow_forwardThe carbonic acid equilibrium is shown below. Exhalation of CO2 by the lungs causes this equilibrium to shift to the ______, which causes the pH of the blood to _______. H+ + HCO3- <=> H2CO3 <=> H2O + CO2 a) left; increase b) left; decrease c) right; decrease d) right; increasearrow_forwardMicky Mango is a 64-year-old male admitted to the emergency room for asthma. His laboratory results are as follows: pH 7.31, pCO2 higher than normal, and total HCO3– also higher than normal. Classify his acid-base balance as acidosis or alkalosis, and as metabolic or respiratory. Is there evidence of compensation? Propose the mechanism by which asthma contributed to the lab results seen.arrow_forward
- Select the best answer or answers from the choices given: A deficiency in this element can be expected to reduce the hemoglobin content of blood: (a) Fe, (b) I, (c) F, (d) Ca, (e) K.arrow_forwardCalculate the hydrogen ion concentration, [H+], for each of the following materials: (a) Blood plasma, pH 7.4 (b) Orange juice, pH 3.5 (c) Human urine, pH 6.2 (d) Household ammonia, pH 11.5 (e) Gastric juice, pH 1.8arrow_forwardHow does HCO3- (Bicarbonate ion) help to maintain the pH balance of the blood?arrow_forward
- The phosphate buffer system is most important in the Choose from the following: (A) respiratory system (B) cardiovascular system (C) digestive system (D) urinary systemarrow_forwardHelen is a 62-year-old smoker. Her physician has diagnosed her as having emphysema that has caused her to hypoventilate. Surprisingly, when her blood pH is checked, it is 7.40. Why?arrow_forwardConsider the equilibrium shown below; indicating the buffering system in the blood circulation. CO, + H,0 [H,CO3] HCO3¯+H* When a patient is choking, would this lead to an alkalosis or an acidosis situation? Explain.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning
Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning

Human Physiology: From Cells to Systems (MindTap ...
Biology
ISBN:9781285866932
Author:Lauralee Sherwood
Publisher:Cengage Learning

Biochemical Tests-Part 1; Author: Southern Stacker;https://www.youtube.com/watch?v=a-i9vANfQWQ;License: Standard Youtube License