
Concept explainers
Interpretation: To identify the site where the enzyme that converts disaccharides to monosaccharides is present.
Concept introduction: Carbohydrates are the
Monosaccharides are the simplest carbohydrate units that cannot be hydrolyzed further to give the smallest units. Disaccharides contain two monosaccharide units and on hydrolysis, it gives two monosaccharide units.
In the digestion process, the food molecules break down into small chemical units through hydrolysis. These small chemical units are used by cells present in the body for their
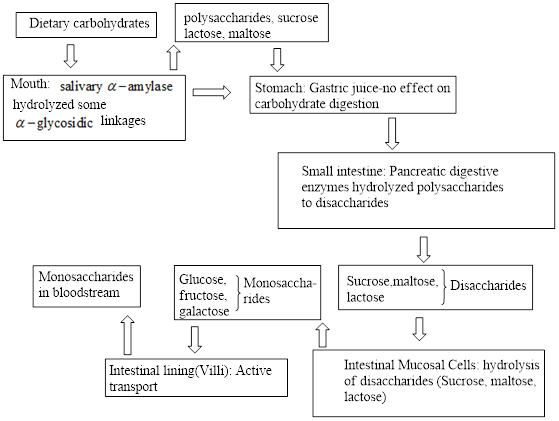
Enzymes are specialized proteins that act as biological catalysts. Enzymes accelerate the biochemical reactions to produce the substances that are needed for cells for their proper functioning.
Want to see the full answer?
Check out a sample textbook solution
Chapter 24 Solutions
General, Organic, and Biological Chemistry
- Question 27 он CHOH он CHOH HO I ACNH II он CH3 OH HO II но What is the glycosidic linkage between monosaccharide Il and III? O B-(1–3) O a-(1-3) O a-(1–2) В-(1—2)arrow_forward6) The disaccharide shown is a но но. HO OH но H. OH B) non reducing sugar A) reducing sugar 7) The glycosidic bond in the following structure can be described as a: OH но. но он OHI B) B (1.1) C) a (14) D) B (14) E) a (2,1) A) a (1,1)arrow_forwardDraw the structure of the disaccharide with a systematic name of: (16)-B-D glucopyranosyl-B-D glucopyranosearrow_forward
- Question 28 CHOH CHOH A CHOH CHOH 1 CH2 CHOH OH OH OH CH 3 CHOH OH OH он CH он он B 2 OH CH Which lettered subunit is the reducing end? O A В There is more than one reducing end on this sugar structurearrow_forward30. Relative specificity means that an enzyme can catalyze a reaction with a. only one specific substance b. structurally related substances C. only D-enantiomers d. no correct responsearrow_forwardThe glycosidic bond in the following structure can be described as a: но- HO. но OH но H H. H. ÓH A ) α ( 1 1) B) B (1,1) C) a (1,4) D) B (1,4) E) a (2,1) F) B (2,1)arrow_forward
- CYCLIZATION OF MONOSACCHARIDESChange the following to Haworth Formula:1. D-idose at C4 ( alpha anomer )2. L- galactose at C5 ( beta anomer )3. D-altrose at C4 ( alpha anomer )4. L-mannose at C5 ( beta anomer )5. D-tallose at C4 ( alpha anomer )arrow_forwardIdentify the isotopes that would be found in each metabolite when [2-¹4C, ¹5N]glutamate undergoes oxidative degradation in the liver of a rat. urea succinate arginine citrulline ornithine aspartate 000000 Answer Bank 15 N 14C I COO- H-15N+- -14℃ H H _________ CH₂ COO- Labeled glutamatearrow_forwardHO 15. (a) Identify the glycosidic bond in the following disaccharide. (b) Decide whether the compound is a non-reducing or reducing sugar. (c) Polysaccharide units are usually bonded together with a or 3 1, 6 or 1, 4 linkages. What linkage is used in the disaccharide shown below? (pis) но- HO HO H HO H HO CHO O 16. Draw the structure for 1,4-ß-D-galactopyranosyl-D-glucose. -OH -OH H но- 17. Assign an R/S designation to each chirality center in the following compound: ( CH₂OH HO O -OHarrow_forward
- QUESTION 24 Given OH HOH,C CHOH OH Which of the following statements are TRUE about the sugar shown above? Litis an L-sugar. IL. It is a D-sugar. Jt is an aldohexose. IV It is a ketohexose V It is an alpha anomer. VI It is a beta-anomer. OLVI O I, II, V OIVV O I, IV VI OIV VI Which one of the following is a possible Haworth structure (alpha or beta) for the monosaccharide shown below CHO HO- CH OH O- CHOH CHOH CH OH CHOH CH CHO OH OH OH O CHO CHOarrow_forwardLIsten What is the FIRST product in the oxidation of fatty acids? O carbon dioxide a caroxylic acid an aldehyde a ketone P Type here to search prt sc delete home & %24 4 backspayce 6 3 Y Q / W / E KLE enter D G J pause M alt ctrl altarrow_forward9) When D-glucose cylclizes to its alpha pyranose form, how many axial hydrogens does it have? a) 3 b) 4 c) 5 d) 6 10) Which blood group has the fewest linked monosaccharides in its antigenic determinant? a) O b) AB c) B d) Aarrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning



