
Concept explainers
Interpretation:
The name of the organic compounds should be given.
Concept introduction:
Organic compounds are named systematically by using IUPAC rules.
Name of the organic compounds are given according to the number of carbon present in the molecule for example
A molecule having one carbon atom, the molecule name will start with meth etc.…

If any halogens are present in the molecule, the name of the halogens as follows.

Naming the substituted
- (1) Name the parent alkane (long alkyl chain)
- (2) Number the carbon
- (3) Name and number the substituent
If the molecules have the multiple substituents, the compound named as di, tri, tetra, penta, ect.
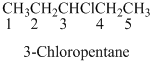
If the molecules having
The given compound is an alcohol
Example is given below
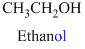

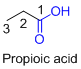
The given compound is an acid (
The amides are derivatives of acids and it is named as the ending of alkane with amide.
For example
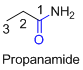
If the molecule is ester,
Esters end with “ate”
Example

The given compound is an
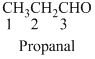
The given compound is
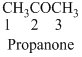
The given compound is an
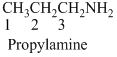
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
Chemistry: Atoms First
- Can you draw this with line structure? (a) trimethylamine (b) 1,4-hexanediamine (c) N,N-dibutylamine (d) 2-Methyl-N-propyl-4-octanaminearrow_forwardGive a chemical test to distinguish between each of the following pairs of compounds :(i) Ethylamine and Aniline(ii) Aniline and Benzylaminearrow_forwardWrite the chemical equation for the reaction of N,N-diethyl-1-propanamine with water. Which is the formula of this rule of reaction? (Hint: the amine is a base):arrow_forward
- 10)2-chloro propanaminearrow_forwardDraw a structural formula for each amine. (a) 1,5-Pentanediamine (b) 2-Bromoaniline (c) Tributylaminearrow_forwardDraw and name compounds that meet these descriptions:(a) Three different amides with the formula C5H11NO(b) Three different esters with the formula C6H12O2arrow_forward
- Write the structures of the following compounds. a) 2-propanamine b) 1-N,N-dimethylaminocyclohexene c) ethanoylbenzoate d) N-methyl-o-toluidine e) 3-Benzyl-5-ethylheptanoic acidarrow_forwardDraw structures corresponding to these names:(a) 4-Methylpentanamide (b) N-Ethyl-N-methylpropanamidearrow_forwardFollowing are structural formulas for two enamines. Me Me క (a) (b) Draw structural formulas for the secondary amine and carbonyl compound from which each enamine is derived.arrow_forward
- Draw the condensed formula or skeletal structural formula for the following compounds: (a) 2,3-dimethylpentanoic acid (b) Ethyl pentanoate (c) N,N-dimethyl-1-propanaminearrow_forwardTRUE OR FALSE (a) There are three amines with the molecular formula C3H9N. (b) Aldehydes, ketones, carboxylic acids, and esters all contain a carbonyl group. (c) A compound with the molecular formula of C3H6O may be either an aldehyde, a ketone, or a carboxylic acid. (d) Bond angles about the carbonyl carbon of an aldehyde, a ketone, a carboxylic acid, and an ester are all approximately 109.5°. (e) The molecular formula of the smallest aldehyde is C3H6O, and that of the smallest ketone is also C3H6O. (f) The molecular formula of the smallest carboxylic acid is C2H4O2.arrow_forwardGive the chemical tests to distinguish between the following pairs of compounds :(i) Ethyl amine and Aniline(ii) Aniline and Benzylaminearrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
