
(a)
Interpretation:
Classification of the reaction should be identified for the given transformation.
Concept introduction:
Substitution reaction: During a
Nucleophilic Substitution reaction: electron rich nucleophile attacks the positive or partially positive charge of an atom and replace a leaving group is called Nucleophilic Substitution reaction.
Elimination reaction: An elimination reaction is removal of two substituents in a molecule and forms
Addition reaction: addition reaction is two or more molecules combine to form a larger one.
Isomerization: one molecule is transformed into another molecule with same molecular formula but different arrangement.
(a)
Answer to Problem 23.52QP
The given reaction is isomerization reaction, the reaction is shown below (a)
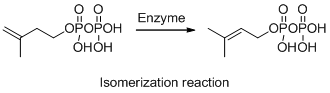
Explanation of Solution
To find:type of the reaction.
The given reaction is isomerization reaction, the reaction is shown below
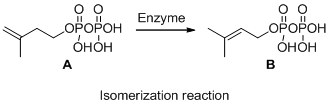
Compound (A) is transformed into compound (B) with same molecular formula but different arrangement so it is called isomerization. The reaction is called isomerization.
(b)
Interpretation:
Classification of the reaction should be identified for the given transformation.
Concept introduction:
Substitution reaction: During a chemical reaction when one functional group is transformed as another functional group in a chemical compound is called substitution reaction.
Nucleophilic Substitution reaction: electron rich nucleophile attacks the positive or partially positive charge of an atom and replace a leaving group is called Nucleophilic Substitution reaction.
Elimination reaction: An elimination reaction is removal of two substituents in a molecule and forms alkene. An elimination reaction is one or two-step process which based on the mechanism when two substituents removed from the molecule in single step is called E1 reaction. When two substituents are removed from the molecule in two steps is called E2 reaction.
Addition reaction: addition reaction is two or more molecules combine to form a larger one.
Isomerization: one molecule is transformed into another molecule with same molecular formula but different arrangement.
(b)
Answer to Problem 23.52QP
The given reaction is substitution reaction, the reaction is shown below (b)
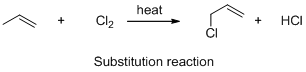
Explanation of Solution
To find: type of the reaction.
The given reaction is substitution reaction, the reaction is shown below
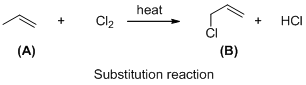
Compound (A) is undergoes reaction with chlorine which forms chlorinatedcompound (B) with elimination of hydrochloric acid in the presence of heat and the hydrogen atom is replaced by the chlorine atom. The reaction is called substitution reaction.
(c)
Interpretation:
Classification of the reaction should be identified for the given transformation.
Concept introduction:
Substitution reaction: During a chemical reaction when one functional group is transformed as another functional group in a chemical compound is called substitution reaction.
Nucleophilic Substitution reaction: electron rich nucleophile attacks the positive or partially positive charge of an atom and replace a leaving group is called Nucleophilic Substitution reaction.
Elimination reaction: An elimination reaction is removal of two substituents in a molecule and forms alkene. An elimination reaction is one or two-step process which based on the mechanism when two substituents removed from the molecule in single step is called E1 reaction. When two substituents are removed from the molecule in two steps is called E2 reaction.
Addition reaction: addition reaction is two or more molecules combine to form a larger one.
Isomerization: one molecule is transformed into another molecule with same molecular formula but different arrangement.
(c)
Answer to Problem 23.52QP
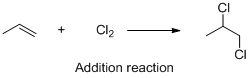
The given reaction is addition reaction, the reaction is shown below (c)
Explanation of Solution
To find: type of the reaction.
The given reaction is addition reaction, the reaction is shown below
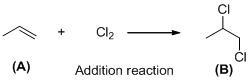
Compound (A) is undergoes addition reaction with chlorine which forms chlorinated compound (B). The reaction is called addition reaction.
(d)
Interpretation:
Classification of the reaction should be identified for the given transformation.
Concept introduction:
Substitution reaction: During a chemical reaction when one functional group is transformed as another functional group in a chemical compound is called substitution reaction.
Nucleophilic Substitution reaction: electron rich nucleophile attacks the positive or partially positive charge of an atom and replace a leaving group is called Nucleophilic Substitution reaction.
Elimination reaction: An elimination reaction is removal of two substituents in a molecule and forms alkene. An elimination reaction is one or two-step process which based on the mechanism when two substituents removed from the molecule in single step is called E1 reaction. When two substituents are removed from the molecule in two steps is called E2 reaction.
Addition reaction: addition reaction is two or more molecules combine to form a larger one.
Isomerization: one molecule is transformed into another molecule with same molecular formula but different arrangement.
(d)
Answer to Problem 23.52QP
The given reaction is substitution reaction, the reaction is shown below (d)
Explanation of Solution
To find: type of the reaction.
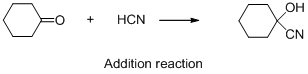
The given reaction is addition reaction, the reaction is shown below (e)
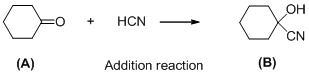
Compound (A) is undergoes reaction with hydrogen cyanide which forms cyanohydrin (B). The reaction is called addition reaction.
(e)
Interpretation:
Classification of the reaction should be identified for the given transformation.
Concept introduction:
Substitution reaction: During a chemical reaction when one functional group is transformed as another functional group in a chemical compound is called substitution reaction.
Nucleophilic Substitution reaction: electron rich nucleophile attacks the positive or partially positive charge of an atom and replace a leaving group is called Nucleophilic Substitution reaction.
Elimination reaction: An elimination reaction is removal of two substituents in a molecule and forms alkene. An elimination reaction is one or two-step process which based on the mechanism when two substituents removed from the molecule in single step is called E1 reaction. When two substituents are removed from the molecule in two steps is called E2 reaction.
Addition reaction: addition reaction is two or more molecules combine to form a larger one.
Isomerization: one molecule is transformed into another molecule with same molecular formula but different arrangement.
(e)
Answer to Problem 23.52QP
The given reaction is addition reaction, the reaction is shown below (e)

The given reaction is elimination reaction, the reaction is shown below (e)
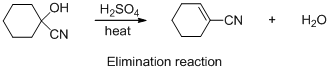
Explanation of Solution
To find: type of the reaction.
The given reaction is elimination reaction, the reaction is shown below
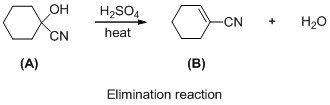
Compound (A) is undergoes elimination reaction with sulfuric acid which forms corresponding elimination compound (B). The reaction is called elimination reaction.
Want to see more full solutions like this?
Chapter 23 Solutions
Chemistry: Atoms First
- Phenylethylamine is a natural substance that is structurally similar to amphetamine. It is found in sources as diverse as almond oil and human urine, where it occurs at elevated concentrations as a result of stress and certain forms of schizophrenia.One method of synthesizing the compound for pharmacological and psychiatric studies involves two steps: Classify each step as an addition, elimination, or substitution.arrow_forwardClassify the following reaction as: addition, elimination, substitution, or rearrangement.arrow_forwardPropene and benzene react with bromine, Br2, in absence of UV light in different ways. The reaction of propene is an addition reaction whereas that of benzene is a substitution reaction. Give the best explanation of this difference.arrow_forward
- Write down the chemical structure of tetrachloroethylene. What type of organic chemical is this (aliphatic or aromatic or cyclic).arrow_forwardOne test for the presence of an alkene is to add a small amount of bromine, which is a red-brown liquid, and look for the disappearance of the red-brown color. This test does not work for detecting the presence of an aromatic hydrocarbon. Explain.arrow_forwardFill in the blanks to complete the following statement. One method used to differentiate between saturated and unsaturated compounds is to add a few drops of orange-coloured aqueous bromine to samples of each organic compound. If the compound is → the orange colour will quickly fade. saturated unsaturated kely reaction for each of the following:arrow_forward
- Write structure of the following compounds= 5-isopropyl-3,3,4-trimethyloctanearrow_forward16. An atom or group of atoms that can give organic compounds distinct chemical and physical properties. 21. When a compound with the general formula R-COOH loses a proton, the product that remains is described with this term. Its general formula is R-COO- 24. A class of organic compounds in which three or more carbons form a ring structure. All of the carbon-to-carbon bonds are single bonds in this family of compounds.arrow_forwardWrite the structure of the compound that will be produced in the following reaction? CH3 –C ≡ C–CH2– CH2 – CH3 + 2HBr→ a) Other than the combustion of alkanes, why are alkanes not reactive, or when they do react, they do so very slowly? B) What chemical reaction can alkenes and alkynes undergo that alkanes cannot? Explainarrow_forward
- Explain why benzene can not undergo electrophilic addition reaction, but can only do electrophilic substitution reaction?arrow_forwardhow do oxidation and reductions correlate with addition, elimination, and substitution reactions?arrow_forwardIdentify the class of organic compound below (alcohol, carboxylic acid, halocarbon, alcohol, ketone, ester, ether). Some words may be used more than oncearrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

