
Concept explainers
(a)
Interpretation:
The structure of the organic compounds should be given.
Concept introduction:
Organic compounds are named systematically by using IUPAC rules.
Name of the organic compounds are given according to the number of carbon present in the molecule for example
A molecule having one carbon atom, the molecule name will start with meth etc.…

If any halogens are present in the molecule, the name of the halogens as follows.

Naming the substituted
- (1) Name the parent alkane (long alkyl chain)
- (2) Number the carbon
- (3) Name and number the substituent
If the molecules have the multiple substituents, the compound named as di, tri, tetra, penta, ect.
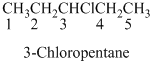
If the molecules having
The given compound is an alcohol
Example is given below
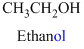
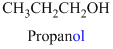
The given compound is an acid (
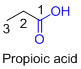
The amides are derivatives of acids and it is named as the ending of alkane with amide.
For example
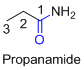
If the molecule is ester,
Esters end with “ate”
Example
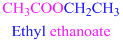
The given compound is an
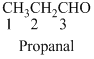
The given compound is an aldehyde (
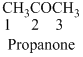
The given compound is an aldehyde (
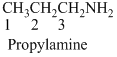
(a)
Answer to Problem 23.20QP
Answer
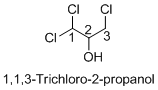
Structure of the given organic compounds is shown below (a).
Explanation of Solution
To find: The structure of the given organic molecule.
Name of the given organic compounds is 1, 1, 3-trichloro-2-propanol.
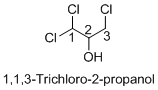
Parent chain is identified and numbering is given for the compound. According to the name, first carbon is bearing two chlorine groups and third carbon is bearing one chlorine group and second carbon is bearing hydroxyl group. The given name is propane which clearly indicates that the compound having three carbon atoms in the molecule. According to the name, the structure of the molecule is given above.
(b)
Interpretation:
The structure of the organic compounds should be given.
Concept introduction:
Organic compounds are named systematically by using IUPAC rules.
Name of the organic compounds are given according to the number of carbon present in the molecule for example
A molecule having one carbon atom, the molecule name will start with meth etc.…

If any halogens are present in the molecule, the name of the halogens as follows.

Naming the substituted alkane:
- (4) Name the parent alkane (long alkyl chain)
- (5) Number the carbon
- (6) Name and number the substituent
If the molecules have the multiple substituents, the compound named as di, tri, tetra, penta, ect.
If the molecules having functional group, the name of the compound is given below. Numbering should be starts from the functional group of the given molecule.
The given compound is an alcohol
Example is given below
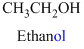
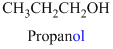
The given compound is an acid (

The amides are derivatives of acids and it is named as the ending of alkane with amide.
For example
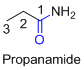
If the molecule is ester,
Esters end with “ate”
Example
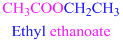
The given compound is an aldehyde (
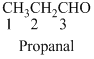
The given compound is an aldehyde (
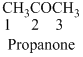
The given compound is an aldehyde (
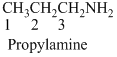
(b)
Answer to Problem 23.20QP
Answer
Structure of the given organic compounds is shown below (b).
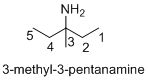
Explanation of Solution
To find: The structure of the given organic molecule.
Name of the given organic compounds is 3-methyl-3-pentanamine.
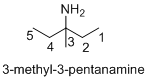
Parent chain is identified and numbering is given for the compound. According to the name, third carbon is bearing methyl group and amine group and the parent name of the molecule is pentane which clearly indicates that the compound having five carbon atoms in the parent. According to the name, the structure of the molecule is given above.
(c)
Interpretation:
The structure of the organic compounds should be given.
Concept introduction:
Organic compounds are named systematically by using IUPAC rules.
Name of the organic compounds are given according to the number of carbon present in the molecule for example
A molecule having one carbon atom, the molecule name will start with meth etc.…

If any halogens are present in the molecule, the name of the halogens as follows.

Naming the substituted alkane:
- (7) Name the parent alkane (long alkyl chain)
- (8) Number the carbon
- (9) Name and number the substituent
If the molecules have the multiple substituents, the compound named as di, tri, tetra, penta, ect.
If the molecules having functional group, the name of the compound is given below. Numbering should be starts from the functional group of the given molecule.
The given compound is an alcohol
Example is given below
The given compound is an acid (

The amides are derivatives of acids and it is named as the ending of alkane with amide.
For example
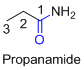
If the molecule is ester,
Esters end with “ate”
Example
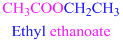
The given compound is an aldehyde (
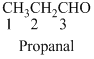
The given compound is an aldehyde (
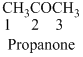
The given compound is an aldehyde (
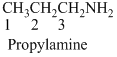
(c)
Answer to Problem 23.20QP
Structure of the given organic compounds is shown below (c).
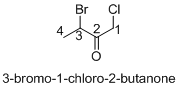
Explanation of Solution
To find: The structure of the given organic molecule.
Name of the given organic compounds is 3-bromo-1-chloro-2-butanone.
Parent chain is identified and numbering is given for the compound. According to the name, first carbon is bearing chlorine atom and third carbon is bearing bromine atom, second carbon is carbonyl carbon and the parent name of the molecule is butane which clearly indicates that the compound having four carbon atoms in the parent. According to the name, the structure of the molecule is given above.
(d)
Interpretation:
The structure of the organic compounds should be given.
Concept introduction:
Organic compounds are named systematically by using IUPAC rules.
Name of the organic compounds are given according to the number of carbon present in the molecule for example
A molecule having one carbon atom, the molecule name will start with meth etc.…

If any halogens are present in the molecule, the name of the halogens as follows.

Naming the substituted alkane:
- (10) Name the parent alkane (long alkyl chain)
- (11) Number the carbon
- (12) Name and number the substituent
If the molecules have the multiple substituents, the compound named as di, tri, tetra, penta, ect.
If the molecules having functional group, the name of the compound is given below. Numbering should be starts from the functional group of the given molecule.
The given compound is an alcohol
Example is given below
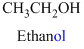
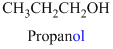
The given compound is an acid (

The amides are derivatives of acids and it is named as the ending of alkane with amide.
For example
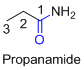
If the molecule is ester,
Esters end with “ate”
Example
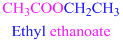
The given compound is an aldehyde (
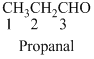
The given compound is an aldehyde (
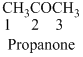
The given compound is an aldehyde (
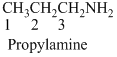
(d)
Answer to Problem 23.20QP
Structure of the given organic compounds is shown below (d).
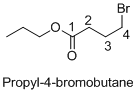
Explanation of Solution
To find: The structure of the given organic molecule.
Name of the given organic compounds is propyl-4-bromobutanoate.
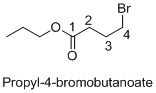
Parent chain is identified and numbering is given for the compound. According to the name, fourth carbon is bearing bromine atom, the given molecule is ester so propyl group is attached after oxygen atom in the molecule and the parent name of the molecule is butane which clearly indicates that the compound having four carbon atoms in the parent. According to the name, the structure of the molecule is given above.
Want to see more full solutions like this?
Chapter 23 Solutions
Chemistry: Atoms First
- Draw the organic product formed when the following compounds undergo a substitution reaction: (a) acetic acid and methyl-amine; (b) butanoic acid and 2-propanol; (c) Formica acid and 2-methyl-1-propanol.arrow_forwardDraw the organic product formed when the following compounds undergo a substitution reaction:(a) acetic and 1-hexanol; (b) propanoic acid and dimethylamine; (c) ethanoic acid and dimethylamine.arrow_forwardDraw the organic product formed when the following compounds undergo a substitution reaction: (a) acetic acid and 1-hexanol; (b) propanoic acid and dimethyl-amine; (c) ethanoic acid and diethylamine.arrow_forward
- Draw the organic product formed when the following compounds undergo a substitution reaction: (a) acetic acid and methylamine; (b) butanoic acid and 2-propanol; (c) formic acid and 2-methyl-1-propanol.arrow_forward5.Write the structural formula of the ester that, when hydrolyzed, would yield the following:(a) methanol and propanoic acid(b) 1-octanol and acetic acid (c) ethanol and butanoic acidarrow_forwardDraw the condensed structure of the compounds formedby condensation reactions between (a) benzoic acid andethanol, (b) ethanoic acid and methylamine, (c) acetic acidand phenol. Name the compound in each case.arrow_forward
- Define ketones, acid halides, esters, amides, and carboxylic acids ?arrow_forwardIdentify which of the following statement(s) is/are true. (f) Aldehydes, ketones, carboxylic acids, and esters all contain a carbonyl group. (g) A compound with the molecular formula of C3H6O may be either an aldehyde, a ketone, or a carboxylic acid. (h) Bond angles about the carbonyl carbon of an aldehyde, a ketone, a carboxylic acid, and an ester are all approximately 109.5°. (i) The molecular formula of the smallest aldehyde is C3H6O, and that of the smallest ketone is also C3H6O. (j) The molecular formula of the smallest carboxylic acid is C2H4O2.arrow_forwardWrite a complete chemical equation showing reactants, products, and catalysts needed (if any) for the following reaction and (2) Draw and name the organic compound found in every reaction.(Use condensed structural formula) (A) Ozonolysis of 3,3-Dimethyloct-4-yne(B) Complete halogenation (Cl2) of 3-Ethyl-5-methyl-1,6,8-decatriyne(C) Partial hydrogenation using Lindlar’s Catalyst 2,2,5,5-Tetramethylhex-3-ynearrow_forward
- Draw complete structural diagrams of the following compounds. (a) 2-methyl,pent-2-ene (b) propyne (c) 2,3-dimethyl butane (d) cycloheptanearrow_forwardWrite both condensed and line structures for (a) the ester formed when butyric acid reacts with cyclopentanol, (b) the amide formed when isopropyl amine is reacted with butyric acid, and (c) the amide formed when diethylamine is reacted with butyric acid.arrow_forwardButanone is a four carbon compound with the functional group:(a) carboxylic acid(b) aldehyde(c) ketone(d) alcoholarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





