
Concept explainers
(a)
Interpretation:
The sample that contains more monounsaturated fatty acids has to be identified.
Concept Introduction:
Monounsaturated fatty acid:
The compound contains only one carbon-carbon double bond in the fatty acid is called as monounsaturated fatty acid.
Example:

(b)
Interpretation:
The sample that contains more polyunsaturated fatty acids has to be identified.
Concept Introduction:
Polyunsaturated fatty acid:
The compound contains more than one carbon-carbon double bond in the fatty acid is called as polyunsaturated fatty acid.
Example:
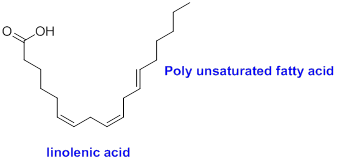
(c)
Interpretation:
The sample that contains fewer trans-fatty acids has to be identified.
Concept Introduction:
Trans-fats:
Trans-fats are the unsaturated fatty acid which occurs in very small amounts in nature, but can be prepared using hydrogenation reaction of vegetable oils.
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- Triacylglycerol A contains 53% oleic acid, 29% linoleic acid, 9% palmitic acid, 4% stearic acid, 1% myristic acid and lesser amount of other fatty acids. Triacylglycerol B contains 39% oleic acid, 29% palmitic acid, 24% stearic acid, 2% myristic acid, 2% linoleic acid and lesser amounts of other fatty acids. Which of the two samples is expected to be solid at room temperature? Explain why.arrow_forwardWhen a 23 Carbon fatty acid is metabolized, what is the total number of (GROSS) ATP produced ?arrow_forwardThe following link carbohydrate metabolism with lipid biosynthesis:(a) How many molecules of glucose are required to provide the carbon forarrow_forward
- For each fatty acid below, categorize it via lipid numbers (e.g. 16:0, 18:1cA9) and, when appropriate, N-x designations (e.g. N-3) a) b) d) MOarrow_forwardWhich of the following represents a proper designation for monounsaturated fatty acid ? 18:0 18:1 (A6) O 17:3(A6,9,12) 17:2 (A5,8)arrow_forwardFatty acids are stored in adipose tissue, as triacylglycerol (TAG) forms. TAGs are degraded as glycerol and fatty acids where energy is required. Based on that knowledge explain: a) How these products are used in the adipose, liver, and other tissue? b)Why glycerol can not be metabolized in adipose tissue?arrow_forward
- Recently it has been suggested that using oils with more monounsaturated fatty acids (e.g., oleic acid) is better for our health than those with polyunsaturated fatty acids or saturated fatty acids. What are good sources of oils with predominantly monounsaturated fatty acids? (Hint: See Table 23.2.)arrow_forward(a) What is protein turnover? Give 1-2 examples. (b) What are the main differences between glucogenic and ketogenic amino acids?arrow_forwardList the classification of fatty acids based on their chemical structures, as well as their synthesis and acquisition from natural food sources. Discuss how they are classified in this manner.arrow_forward
- Describe the biological significance of cholesterol.arrow_forwardWhich of these fatty acids has the lower melting point? Explain why.(a) Linoleic acid (b) Stearic acidarrow_forward1) An average middle-aged person weighing 90 kg (200 lb.) contains 15 % body fat stored in adipose tissue. a) Calculate the amount of energy stored as fat in this person in kilojoules, assuming that energy yield from fat is 37 kJ/g. b) If this same amount of energy were stored in hydrated glycogen rather than fat (hydrated glycogen has an energy yield of 6 kJ/g), how much would the person weight in kilograms and in pounds?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





