
Organic Chemistry (8th Edition)
8th Edition
ISBN: 9780134042282
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
thumb_up100%
Chapter 22.9, Problem 15P
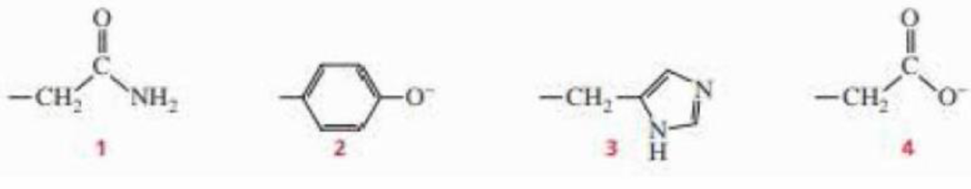
Which of the following amino acid side chains can help remove a proton from the α-carbon of an
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which of the following amino acid side chains can aid the departure of a leaving group?
Which of the following amino acid's side chains will become protonated and carry a +1 charge when the pH of its environment is decreased from pH 11.5 to pH 7.5?
Glutamate
Lysine
Arginine
Histidine
Aminotransferases require Mg2+ ion as a cofactor.True or False?
Chapter 22 Solutions
Organic Chemistry (8th Edition)
Ch. 22.2 - Compare each of the mechanisms listed here with...Ch. 22.2 - Prob. 3PCh. 22.2 - Prob. 4PCh. 22.3 - a. Draw the mechanism for the following reaction...Ch. 22.5 - Prob. 7PCh. 22.5 - Propose a mechanism for the Co2+ catalyzed...Ch. 22.6 - Prob. 9PCh. 22.7 - Prob. 10PCh. 22.7 - Prob. 12PCh. 22.7 - Prob. 13P
Ch. 22.9 - Which of the following amino acid side chains can...Ch. 22.9 - Which of the following C-terminal peptide bonds is...Ch. 22.9 - Carboxypeptidase A has esterase activity as well...Ch. 22.10 - Arginine and lysine side chains fit into trypsins...Ch. 22.10 - Explain why serine proteases do not catalyze...Ch. 22.11 - If H2 18O is used in the hydrolysis reaction...Ch. 22.11 - Draw the pH-activity profile for an enzyme that...Ch. 22.12 - The pHactivity profile for glucose-6-phosphate...Ch. 22.12 - Prob. 23PCh. 22.13 - Draw the mechanism for the hydroxide ion-catalyzed...Ch. 22.13 - What advantage does the enzyme gain by forming an...Ch. 22.13 - Prob. 26PCh. 22.13 - Prob. 27PCh. 22.13 - Aldolase shows no activity if it is incubated with...Ch. 22 - Which of the following parameters would be...Ch. 22 - Prob. 29PCh. 22 - Prob. 30PCh. 22 - Prob. 31PCh. 22 - Indicate the type of catalysis that is occurring...Ch. 22 - The deuterium kinetic isotope effect (KH2O/KD2O)...Ch. 22 - Prob. 34PCh. 22 - Co2+ catalyzes the hydrolysis of the lactam shown...Ch. 22 - there are two kinds of aldolases. Class I...Ch. 22 - Prob. 37PCh. 22 - The hydrolysis of the ester shown here is...Ch. 22 - Prob. 39PCh. 22 - At pH = 12, the rate of hydrolysis of ester A is...Ch. 22 - 2-Acetoxycyclohexyl tosylate reacts with acetate...Ch. 22 - Proof that an imine was formed between aldolase...Ch. 22 - Prob. 43PCh. 22 - a. Explain why the alkyl halide shown here reacts...Ch. 22 - Triosephosphate isomerase (TIM) catalyzes the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 22-71 Which amino acid side chain is most frequently involved in denaturation by reduction?arrow_forwardAn amino acid residue side chain is deprontonated at pH9.5. What is the pKa of this residue if it is fully deprotonated at this pH?arrow_forwardTyrosine is an amino acid whose side chain has a pKa of 10.1. At pH 7, what protonation form would you expect to find it in?arrow_forward
- Carboxypeptidase requires a Zn²+ cofactor for the hydrolysis of the peptide bond of a C-terminal aromatic amino acid. T/F?arrow_forwardExplain why, when the guanidino group of arginine is protonated, the double-bonded nitrogen is thenitrogen that accepts the proton. 2 H+ + NH2NHCH2CH2CH2CHNH +NH3CO−O CO−O H2NC NHCH2CH2CH2CHarrow_forwardDraw the structure of the amino acid L-leucine at each pH: (a) 6; (b) 10; (c) 2. Which form predominates at L-leucine's isoelectric point?arrow_forward
- What is the predominant form of the following amino acids at pH= 1? What is the overall charge on the amino acid at this pH? threoninearrow_forwardWhat is the predominant form of the following amino acids at pH= 1? What is the overall charge on the amino acid at this pH? argininearrow_forwardwould chymotrypsin to be most active at a pH of 4.5, 7.8, or 10.0?arrow_forward
- Although tryptophan contains a heterocyclic amine, it is considered a neutral aminoacid. Explain why the indole nitrogen of tryptophan is more weakly basic than one ofthe imidazole nitrogens of histidine.arrow_forwardsuggest a plausible arrow-pushing mechanism for the formation of a covalent adduct between the suicide inhibitor 5-trifluoromethyluridylic acid and thymidylate synthase that involves the active_site thiolate.arrow_forwardShow how the following amino acids might be formed in the laboratory by reductiveamination of the appropriate a@ketoacid.(a) alanine (b) leucine (c) serine (d) glutaminearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY