
Organic Chemistry - Standalone book
10th Edition
ISBN: 9780073511214
Author: Francis A Carey Dr., Robert M. Giuliano
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 22, Problem 31P
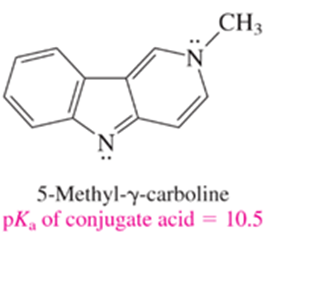
The compound shown is a somewhat stronger base than ammonia. Which nitrogen do you think is protonated when it is treated with an acid? Write a structural formula for the species that results.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
(a) Rank the following compounds in order of increasing acidity. (b) Which compound forms the strongest conjugate base?
Wouldn't the oxygen be protonated since it becomes more electronegative through resonance and more willing to accept a proton?
Which of the two compounds shown below is acidic? Why?
Chapter 22 Solutions
Organic Chemistry - Standalone book
Ch. 22.1 - Prob. 1PCh. 22.1 - Prob. 2PCh. 22.1 - Prob. 3PCh. 22.2 - Prob. 4PCh. 22.4 - Prob. 5PCh. 22.4 - Prob. 6PCh. 22.4 - Prob. 7PCh. 22.4 - Prob. 8PCh. 22.4 - Prob. 9PCh. 22.7 - Prob. 10P
Ch. 22.8 - Problem 22.11 Three of the following amines can be...Ch. 22.9 - Prob. 12PCh. 22.10 - Prob. 13PCh. 22.13 - Prob. 14PCh. 22.14 - Prob. 15PCh. 22.15 - Prob. 16PCh. 22.15 - Prob. 17PCh. 22.17 - Prob. 18PCh. 22.17 - Prob. 19PCh. 22.17 - Prob. 20PCh. 22.17 - Prob. 21PCh. 22.18 - Prob. 22PCh. 22 - Prob. 23PCh. 22 - Prob. 24PCh. 22 - Prob. 25PCh. 22 - Prob. 26PCh. 22 - Prob. 27PCh. 22 - Arrange the following compounds or anions in each...Ch. 22 - Prob. 29PCh. 22 - Prob. 30PCh. 22 - The compound shown is a somewhat stronger base...Ch. 22 - Prob. 32PCh. 22 - Prob. 33PCh. 22 - Prob. 34PCh. 22 - Prob. 35PCh. 22 - Prob. 36PCh. 22 - Write the structure of the product formed on...Ch. 22 - Prob. 38PCh. 22 - Prob. 39PCh. 22 - Prob. 40PCh. 22 - Prob. 41PCh. 22 - Prob. 42PCh. 22 - Prob. 43PCh. 22 - Prob. 44PCh. 22 - Devise efficient syntheses of each of the...Ch. 22 - Prob. 46PCh. 22 - Prob. 47PCh. 22 - Prob. 48PCh. 22 - Prob. 49PCh. 22 - Prob. 50PCh. 22 - Prob. 51PCh. 22 - Prob. 52PCh. 22 - Prob. 53PCh. 22 - Prob. 54PCh. 22 - Prob. 55PCh. 22 - Prob. 56DSPCh. 22 - Prob. 57DSPCh. 22 - Prob. 58DSPCh. 22 - Prob. 59DSPCh. 22 - Synthetic Applications of Enamines The formation...Ch. 22 - Prob. 61DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Compare with explanation the acidity of the following pairs of compounds. (Note: You must draw the appropriate resonance structures to support your answers.) (c) (i) .COOH .COOH H3COarrow_forwardWhich compound in each of the following pairs would have the higher .boiling point? Explain your answers (1) or HO, (a) (b) (2) HO. OH or (a) (Б) (3) -OH or (a) (Ь)arrow_forwardBy drawing the lewis structure of the cyanide anion, interpret according to the molecular orbital theory on which atom the active end is on, that is, which atom it react in a reaction?arrow_forward
- Suggest a possible structure for Compound X.arrow_forwardA carboxylic acid has two oxygen atoms, each with two nonbonding pairs of electrons.(a) Draw the resonance forms of a carboxylic acid that is protonated on the hydroxyoxygen atom.arrow_forwardLike nitrogen and carbon, oxygen also shows this same hybridization effect on acidity. Both of the following compounds can lose a proton from a positively charged oxygen with three bonds to give a conjugate base containing neutral oxygen with two bonds. One of these structures has pKa = -2.4, while the other has pKa = -8.0. Show the reaction of each compound with water.arrow_forward
- Would the carboxyl group make the 3rd option the most acidic? Or would the fluorines make the 5th one the most acidic?arrow_forwardA carboxylic acid has two oxygen atoms, each with two nonbonding pairs of electrons.(a) Draw the resonance forms of a carboxylic acid that is protonated on the hydroxyoxygen atom.(b) Compare the resonance forms with those given previously for an acid protonated on thecarbonyl oxygen atomarrow_forwardRank the following compounds in order of increasing acidity (1 = least acidic, 3 = most acidic) and in the space provided use resonance (of the conjugate base) to explain why the compound you have labelled “3” is the most acidic.arrow_forward
- Compare and explain the acidity of phenol, ethanol, acetic acid. (Using pKa, structures of conjugate base)arrow_forwardYou will not find “hydroxide” in the stockroom, but you will find sodium hydroxide (NaOH) andpotassium hydroxide (KOH). Lithium hydroxide (LiOH) is expensive and used in spacecraft airfilters since hydroxide reacts with carbon dioxide, and lithium is lighter than sodium or potassium.Cesium and francium hydroxides are very expensive and little used. Is this information consistentwith your answer to the previous question?arrow_forwardUsing curved arrows to show movement of electron pairs, show the mechanism for this reaction. It is only necessary to draw one resonance structure for any intermediates that are resonance hybrids. Note that the H atom of the hydroxyl group is acidic enough to be removed by OH–; the first step in the mechanism is a proton transfer. The general mechanism for base-mediated saponification of an ester is outlined in one of the discussion videos that has been posted for this experiment.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY