
CHEMISTRY-TEXT
8th Edition
ISBN: 9780134856230
Author: Robinson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 22, Problem 22.6A
Look at the location of elements A, B, C, and Din the following periodic table: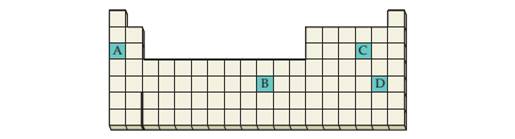
(a) Write the formula of the simplest binary hydride of eachelement.
(b) Classify each binary hydride as ionic, covalent, or interstitial.
(c) Which of these hydrides are molecular? Which are solids withan infinitely extended three-dimensional crystal structure?
(d) What are the oxidation states of hydrogen and the otherelement in the hydrides of A, C, and D?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Write a balanced equation for each of the followingreactions: (a) Burning magnesium metal in a carbondioxide atmosphere reduces the CO2 to carbon. (b) Inphotosynthesis, solar energy is used to produce glucose(C6H12O6) and O2 from carbon dioxide and water.(c) When carbonate salts dissolve in water, they producebasic solutions.
Write a balanced equation for each of the following reactions:(a) Hydrogen cyanide is formed commercially bypassing a mixture of methane, ammonia, and air over acatalyst at 800 °C. Water is a by-product of the reaction.(b) Baking soda reacts with acids to produce carbon dioxidegas. (c) When barium carbonate reacts in air with sulfurdioxide, barium sulfate and carbon dioxide form.
Assign reason for the following :
(i) Sulphur in vapour state exhibits paramagnetism.
(ii) F2 is strongest oxidising agent among halogens.
(iii) Inspite of same electronegativity oxygen forms hydrogen bond while chlorine does not.
Chapter 22 Solutions
CHEMISTRY-TEXT
Ch. 22 - Which element has more nonmetallic character:Cl or...Ch. 22 - Prob. 22.2ACh. 22 - Prob. 22.3PCh. 22 - Prob. 22.4ACh. 22 - Prob. 22.5PCh. 22 - Look at the location of elements A, B, C, and Din...Ch. 22 - What are the formula and charge of the silicate...Ch. 22 - Suggest a plausible structure for the silicate...Ch. 22 - Prob. 22.9PCh. 22 - Write balanced net ionic equations for the...
Ch. 22 - Liquid hydrogen has been used as a fuel in theU.S....Ch. 22 - (a) Write balanced equations for the...Ch. 22 - Write a balanced equation for the production of...Ch. 22 - Prob. 22.14PCh. 22 - Prob. 22.15PCh. 22 - Prob. 22.16PCh. 22 - Prob. 22.17PCh. 22 - Locate each of the following groups of elements on...Ch. 22 - Prob. 22.19CPCh. 22 - Prob. 22.20CPCh. 22 - Prob. 22.21CPCh. 22 - Prob. 22.22CPCh. 22 - Prob. 22.23CPCh. 22 - Prob. 22.24CPCh. 22 - Prob. 22.25CPCh. 22 - Prob. 22.26CPCh. 22 - Prob. 22.27CPCh. 22 - Prob. 22.28CPCh. 22 - Consider the six second- and third-row elements in...Ch. 22 - Prob. 22.30CPCh. 22 - Prob. 22.31CPCh. 22 - Which element in each of the following pairs has...Ch. 22 - Arrange the following elements in order of...Ch. 22 - Prob. 22.34SPCh. 22 - Arrange the following elements in order of...Ch. 22 - Prob. 22.36SPCh. 22 - Arrange the following elements in order of...Ch. 22 - Which element in each of the following pairs has...Ch. 22 - Which element in each of the following pairs has...Ch. 22 - Prob. 22.40SPCh. 22 - Prob. 22.41SPCh. 22 - Prob. 22.42SPCh. 22 - Prob. 22.43SPCh. 22 - Consider the elements C, Se, B, Sn, and Cl....Ch. 22 - Prob. 22.45SPCh. 22 - BF3 reacts with F to give BF4 , but AlF3 reacts...Ch. 22 - GeCl4 reacts with Cl to give GeCl62 , but CCl4...Ch. 22 - At ordinary temperatures, sulfur exists as S8 but...Ch. 22 - Carbon, nitrogen, and oxygen form bonds, but...Ch. 22 - Prob. 22.50SPCh. 22 - Consider the elements Mn, Al, C, S, and Si. Which...Ch. 22 - Prob. 22.52SPCh. 22 - Prob. 22.53SPCh. 22 - Prob. 22.54SPCh. 22 - The hydrogen-filled dirigible Hindenburg had a...Ch. 22 - Write the chemical formula of a compound that...Ch. 22 - Prob. 22.57SPCh. 22 - Prob. 22.58SPCh. 22 - Prob. 22.59SPCh. 22 - Prob. 22.60SPCh. 22 - Prob. 22.61SPCh. 22 - Prob. 22.62SPCh. 22 - Describe the molecular geometry of: (a) GeH4(b)...Ch. 22 - Prob. 22.64SPCh. 22 - Explain why the hydrogen atoms in interstitial...Ch. 22 - Write a balanced net ionic equation for the...Ch. 22 - Write a balanced net ionic equation for the...Ch. 22 - Look at the properties of the alkali metals...Ch. 22 - Why does chemical reactivity increase from top to...Ch. 22 - Prob. 22.70SPCh. 22 - Prob. 22.71SPCh. 22 - Prob. 22.72SPCh. 22 - Prob. 22.73SPCh. 22 - Prob. 22.74SPCh. 22 - Prob. 22.75SPCh. 22 - Magnesium metal is produced by electrolysis of...Ch. 22 - How many hours are required to produce 10.0 kg of...Ch. 22 - Assign charges to the oxygen-containing anions in...Ch. 22 - Assign charges to the oxygen-containing anions in...Ch. 22 - Prob. 22.80SPCh. 22 - Prob. 22.81SPCh. 22 - Prob. 22.82SPCh. 22 - What is the oxidation state of the group 3A...Ch. 22 - Prob. 22.84SPCh. 22 - Prob. 22.85SPCh. 22 - Prob. 22.86SPCh. 22 - Prob. 22.87SPCh. 22 - Prob. 22.88SPCh. 22 - Prob. 22.89SPCh. 22 - Prob. 22.90SPCh. 22 - Prob. 22.91SPCh. 22 - Draw the electron-dot structure for CO, CO2 , and...Ch. 22 - What is the hybridization and geometry around...Ch. 22 - Which of the group 4A elements have allot ropes...Ch. 22 - Prob. 22.95SPCh. 22 - Prob. 22.96SPCh. 22 - Prob. 22.97SPCh. 22 - Prob. 22.98SPCh. 22 - Prob. 22.99SPCh. 22 - Prob. 22.100SPCh. 22 - Suggest a plausible structure for the silicate...Ch. 22 - Carbon is an essential element in the molecules on...Ch. 22 - Prob. 22.103SPCh. 22 - Prob. 22.104SPCh. 22 - Prob. 22.105SPCh. 22 - Prob. 22.106SPCh. 22 - Prob. 22.107SPCh. 22 - Draw an electron-dot structure for N2 , and...Ch. 22 - Describe the structures of the white and red...Ch. 22 - Prob. 22.110SPCh. 22 - Prob. 22.111SPCh. 22 - Account for each of the following observations....Ch. 22 - Compare and contrast the properties of ammonia and...Ch. 22 - Draw all the possible resonance structure for N2O...Ch. 22 - Could the strain in the P4 molecule be reduced by...Ch. 22 - Prob. 22.116SPCh. 22 - Prob. 22.117SPCh. 22 - In industry O2 is prepared by fractional...Ch. 22 - Prob. 22.119SPCh. 22 - Prob. 22.120SPCh. 22 - Prob. 22.121SPCh. 22 - Prob. 22.122SPCh. 22 - Prob. 22.123SPCh. 22 - Prob. 22.124SPCh. 22 - Prob. 22.125SPCh. 22 - Prob. 22.126SPCh. 22 - Prob. 22.127SPCh. 22 - Which is more acidic? (a) Cr2O3orCrO3 (b)...Ch. 22 - Prob. 22.129SPCh. 22 - Write a balanced net ionic equation for the...Ch. 22 - Write a balanced net ionic equation for the...Ch. 22 - Prob. 22.132SPCh. 22 - Write a balanced net ionic equation for the...Ch. 22 - Describe the structure of the sulfur molecules in:...Ch. 22 - The viscosity of liquid sulfur increases sharply...Ch. 22 - Prob. 22.136SPCh. 22 - Write a balanced net ionic equation for each of...Ch. 22 - Prob. 22.138SPCh. 22 - Prob. 22.139SPCh. 22 - Write electron-dot structures for each of the...Ch. 22 - (a) Why is the SO3 molecule trigonal planar hut...Ch. 22 - Prob. 22.142SPCh. 22 - Prob. 22.143SPCh. 22 - Little is known about the chemistry of astatine...Ch. 22 - Prob. 22.145SPCh. 22 - Prob. 22.146SPCh. 22 - Prob. 22.147SPCh. 22 - Prob. 22.148SPCh. 22 - Prob. 22.149SPCh. 22 - Prob. 22.150SPCh. 22 - Prob. 22.151SPCh. 22 - Prob. 22.152SPCh. 22 - Prob. 22.153SPCh. 22 - Prob. 22.154SPCh. 22 - Prob. 22.155SPCh. 22 - Prob. 22.156SPCh. 22 - Prob. 22.157SPCh. 22 - Prob. 22.158SPCh. 22 - Prob. 22.159SPCh. 22 - Prob. 22.160MPCh. 22 - Prob. 22.161MPCh. 22 - Prob. 22.162MPCh. 22 - Prob. 22.163MPCh. 22 - Prob. 22.164MPCh. 22 - Prob. 22.165MPCh. 22 - Prob. 22.166MPCh. 22 - Prob. 22.167MP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Assign reason for the following :(i) Sulphur in vapour state exhibits paramagnetism.(ii) F2 is strongest oxidising agent among halogens.(iii) In spite of having same electronegativity, oxygen forms hydrogen bond while chlorine does not.arrow_forward(a) Account for the following: (i) Helium is used in diving apparatus. (ii) Fluorine does not exhibit positive oxidation state. (iii) Oxygen shows catenation behaviour less than sulphur.arrow_forwardWrite the chemical formula for each of the following, andindicate the oxidation state of the halogen or noble-gasatom in each: (a) calcium hypobromite, (b) bromic acid,(c) xenon trioxide, (d) perchlorate ion, (e) iodous acid,(f) iodine pentafluoride.arrow_forward
- Write balanced equations for the thermal decomposition of potassium nitrate (O₂ is also formed in both cases): (a) at lowtemperature to the nitrite; (b) at high temperature to the metaloxide and nitrogen.arrow_forward(a) Mention the optimum conditions for the industrial manufacture of ammonia by Haber’s process. (b) Explain the following giving appropriate reasons: (i) Sulphur vapour exhibits paramagnetic behaviour: (ii) Red phosphorus is less reactive than white phosphorusarrow_forwardDisulfides are compounds that have S— S bonds, like peroxideshave O— O bonds. Thiols are organic compoundsthat have the general formula R— SH, where R is a generichydrocarbon. The SH- ion is the sulfur counterpart ofhydroxide, OH-. Two thiols can react to make a disulfide,R— S— S— R. (a) What is the oxidation state of sulfur ina thiol? (b) What is the oxidation state of sulfur in a disulfide?(c) If you react two thiols to make a disulfide, are youoxidizing or reducing the thiols? (d) If you wanted to converta disulfide to two thiols, should you add a reducingagent or oxidizing agent to the solution? (e) Suggest whathappens to the H’s in the thiols when they form disulfides.arrow_forward
- What are the oxidation numbers of the following: (a) Si in SiO2 (b) N in NH4+ (c) N in NH3 (d) N in N2Oarrow_forwardTable gives the ionic radii for K^+ and O^2- as 0.138 and 0.140nm, respectively.(a) What is the coordination number for each O^2- ion?(b) Brief y describe the resulting crystal structure for K2O.(c) Explain why this is called the antif uorite structurearrow_forwardConsider the following three statements about the reactivityof an alkali metal M with oxygen gas:(i) Based on their positions in the periodic table, the expectedproduct is the ionic oxide M2O.(ii) Some of the alkali metals produce metal peroxides or metalsuperoxides when they react with oxygen.(iii) When dissolved in water, an alkali metal oxide produces abasic solution.Which of the statements (i), (ii), and (iii) is or are true?(a) Only one of the statements is true.(b) Statements (i) and (ii) are true.(c) Statements (i) and (iii) are true.(d) Statements (ii) and (iii) are true.(e) All three statements are true.arrow_forward
- (a) Two unlabelled bottles are suspected to contain beryllium and magnesium. (i) Suggest a simple test to differentiate the metals present in the unlabelled bottles. (ii) State the observation obtained and write a balanced chemical equation for the reaction that takes place.arrow_forwardNitrous acid (HNO2) disproportionates in acidic solutionto nitrate ion (NO3- ) and nitrogen oxide (NO). Write abalanced equation for this reaction.arrow_forwardCompare and contrast the chemistry of group 1 elements with that of group 2 on the basis of the following:(8)a)Nature of oxidesb)Solubility and thermal stability of the carbonatesc)Polarizing power of cationsd)Reactivity and reducing powerarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning
DISTINCTION BETWEEN ADSORPTION AND ABSORPTION; Author: 7activestudio;https://www.youtube.com/watch?v=vbWRuSk-BhE;License: Standard YouTube License, CC-BY
Difference Between Absorption and Adsorption - Surface Chemistry - Chemistry Class 11; Author: Ekeeda;https://www.youtube.com/watch?v=e7Ql2ZElgc0;License: Standard Youtube License