
Concept explainers
(a)
Interpretation:
The individual amino acids in following dipeptide and the name of it using three-letter abbreviation should be determined:
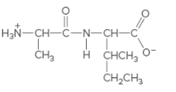
Concept Introduction:
An
(b)
Interpretation:
The individual amino acids in following dipeptide and the name of it using three-letter abbreviation should be determined:
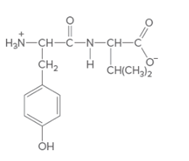
Concept Introduction:
An amine or peptide bond is a chemical bond. It is formed when amino groups of one molecule is reacts with carboxylic group of other molecule. Derivation of two amino acid is known as dipeptide.
Want to see the full answer?
Check out a sample textbook solution
Chapter 21 Solutions
General, Organic, and Biological Chemistry - 4th edition
- What characteristics indicate that amino acids exist as zwitterions?arrow_forwardWhich of the following describes the primary structure of proteins? a. The collective shape assumed by all of the chains in a protein containing multiple chains. b. The folding of an individual protein molecule. c. The regular repeated shape of the protein molecules backbone. d. The sequence of amino acids bonded together by peptide bonds.arrow_forwardAt room temperature, amino acids are solids with relatively high decomposition points. Explain why.arrow_forward
- Consider the tripeptide tyrosylleucylisoleucine. a. Specify its structure using three-letter symbols for the amino acids. b. How many peptide bonds are present within the peptide? c. Which of the amino acid residues has the largest R group? d. Which of the amino acid residues, if any, has an acidic side chain?arrow_forwardIn a pleated sheet secondary structure for a protein a. describe the general shape of the protein backbone b. describe the general locations for the amino acid R groupsarrow_forwardConsider the tripeptide leucylvalyltryptophan. a. Specify its structure using three-letter symbols for the amino acids. b. How many peptide bonds are present within the peptide? c. Which of the amino acid residues has the largest R group? d. Which of the amino acid residues, if any, has a basic side chain?arrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





