
Organic Chemistry (6th Edition)
6th Edition
ISBN: 9781260119107
Author: Janice Gorzynski Smith
Publisher: McGraw Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 21.2, Problem 1P
Draw the enol or keto tautomer(s) of each compound.
a. 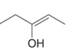 c.
c. 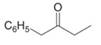 e.
e. 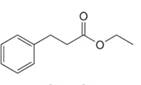
b. 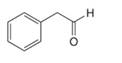 d.
d. 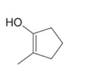 f.
f. 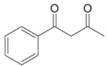 [Draw mono enol
[Draw mono enol
tautomers only]
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Devise a synthesis of each compound from an alkyl halide using any other organic or inorganic reagents.
SH
b.
c. CH;CH,CN
d.
e. CH,CH,OCOCH3
a.
Draw the product formed from a Michael reaction with the given starting materials using OEt and
EtOH.
&. i
CO₂Et
NaOEt
EtOH
draw structure ...
pls draw a stepwise mechanism for the reaction thanks!
Chapter 21 Solutions
Organic Chemistry (6th Edition)
Ch. 21.2 - Problem 23.1 Draw the enol or keto tautomer(s) of...Ch. 21.2 - Problem 23.3 When phenylacetaldehyde is dissolved...Ch. 21.3 - Prob. 5PCh. 21.3 - Problem 23.5 Which bonds in the following...Ch. 21.3 - Prob. 7PCh. 21.3 - Prob. 8PCh. 21.3 - Prob. 9PCh. 21.4 - Prob. 10PCh. 21.5 - Prob. 11PCh. 21.7 - Problem 23.11 Draw the products of each...
Ch. 21.7 - Problem 23.12 Draw the products of each reaction....Ch. 21.7 - Prob. 14PCh. 21.7 - Prob. 15PCh. 21.8 - Prob. 16PCh. 21.8 - Prob. 17PCh. 21.8 - Prob. 18PCh. 21.8 - Problem 23.18 How can pentan-2-one be converted...Ch. 21.8 - Problem 23.19 Identify A, B, and C, intermediates...Ch. 21.9 - Problem 23.20 Which of the following compounds...Ch. 21.9 - Problem 23.21 Draw the products of each...Ch. 21.9 - Prob. 23PCh. 21.9 - Prob. 24PCh. 21.9 - Prob. 25PCh. 21.10 - Prob. 26PCh. 21.10 - Prob. 27PCh. 21.10 - Prob. 28PCh. 21.10 - Prob. 29PCh. 21 - 23.29 Draw enol tautomer(s) for each compound....Ch. 21 - 22.30 The cis ketone A is isomerized to a trans...Ch. 21 - 23.31 Draw enol tautomer(s) for each compound.
...Ch. 21 - Prob. 33PCh. 21 - Prob. 34PCh. 21 - 23.35 Rank the labeled protons in each compound in...Ch. 21 - Prob. 36PCh. 21 - Prob. 37PCh. 21 - 23.38 Acyclovir is an effective antiviral agent...Ch. 21 - 23.39 Explain why forms two different alkylation...Ch. 21 - Prob. 40PCh. 21 - 23.42 Draw a stepwise mechanism for the following...Ch. 21 - Prob. 42PCh. 21 - Prob. 43PCh. 21 - 23.45 Devise a synthesis of valproic acid , a...Ch. 21 - Prob. 57PCh. 21 - 23.57 Draw a stepwise mechanism showing how two...Ch. 21 - 23.58 Draw a stepwise mechanism for the following...Ch. 21 - Prob. 65PCh. 21 - 23.66 Synthesize (Z)-hept-5-en-2-one from ethyl...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- draw all the possiple reagent fir each onearrow_forward16.43 Draw the products of each reaction. a. b. d. HO C. H Lore L sob Br₂ FeBr3 e. NO₂ Br HNO3 H₂SO4 CI AICI 3 Cl₂ FeCl3 SO3 H₂SO4arrow_forwardDraw the product formed when A is treated with each series of reagents.a. [1] H2O; [2] NaH; [3] CH3Brb. [1] CuCN; [2] DIBAL-H; [3] H2O c. [1] C6H5NH2; [2] CH3COClarrow_forward
- 16.41 Draw the products of each reaction. а. HNO3 H2SO4 'NO2 HO b. AICI3 CI С. H. Br2 FeBr3arrow_forward1. Draw the products of each nucleophilic substitution reaction a. b. D b OH C. d. e. f. Br 1 NaCN + NaOCH3 H₂Oarrow_forwardElectrophilic Addition Soubong neblA-aleid rose 9160910 31 babeen ene singo 14.43 Draw the products formed when each compound is treated with one equivalent of HBr. a. b. C.arrow_forward
- Problem 13.34 Synthesize each compound from cyclohexanol, ethanol, and any other needed reagents. a. b. C. d. e. OH XOH Br OHarrow_forwardDraw the enol form of each keto tautomer in parts (a) and (b), and the keto form of each enol tautomer in parts (c) and (d).arrow_forwardIdentify the keto form of each enol tautomer. A. HO. H. B. OH C. HO A1 D. В1 C1 D1 C2 Resetarrow_forward
- Devise a synthesis of each compound from cyclopentane and any other required organic or inorganic reagents. OH C. g. -OH a. .CI Br OH b. -Br d. h. Br Br CNarrow_forwardDraw the keto tautomer of each enol.arrow_forwardWhat is the best reagent to complete this synthesis? 1)???? 2) NaCN HO CN A. PBr, CISIMEB sOCl, MSCI B. C. D.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY