
Concept explainers
The Enolate Chemistry of Dianionss
The synthetic applications of carbanions as reagents for carbon
Consider acetic acid:
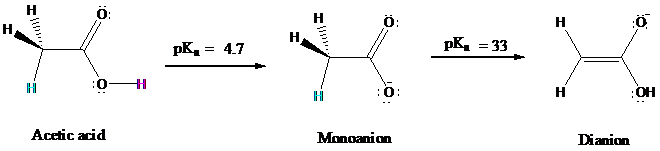
The pKa of acetic acid is
for ionization of a
is shared by carbon. The dianion, however, has carbanionic character and the potential to act as a
nucleophile in carbon
Diisopropylamine has a pKa of
enough base to convert acetic acid to its dianion. Other carboxylic acids behave similarly to give
dianions that undergo typical carbanion reactions. Alkylation of
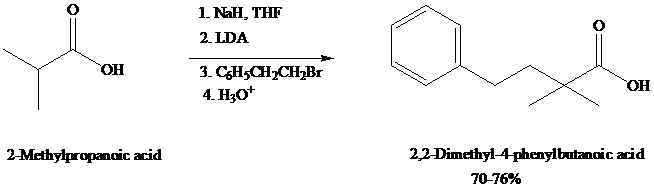
Experimentally, as in this example, it is sometimes useful to convert the carboxylic acid to its carboxylate (monoanion) with sodium hydride (NaH, step
The dianions of α-halocarboxylic acids give epoxy acids (called glycidic acids) on reaction with
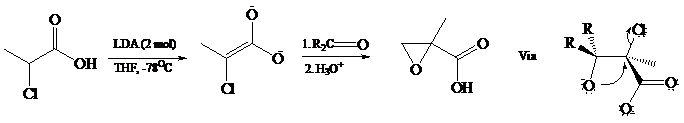
Dianions have been prepared from
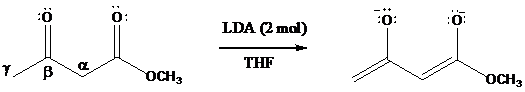
Protons on the
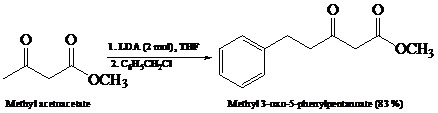
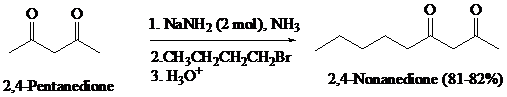
What is the product of the reaction of the dianion in Problem
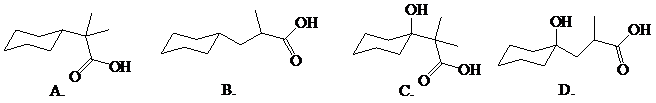
Predict the major organic product(s) of the following reaction

Want to see the full answer?
Check out a sample textbook solution
Chapter 21 Solutions
Organic Chemistry - Standalone book
- Interpret the acidity of alcohols on the basis of ground-state polarization and stability of the alcoholate anion(indicate and give symbols for bond polarization)! Compare the relative acidity of ethanol and 2-fluoroethanol!arrow_forward(a) Tsomane and Nyiko were given a task of synthesising methylenecyclohexane 2. After a brief discussion with each other, Tsomane proposed Method A to synthesise 2 from cyclohexanone 1 while Nyiko proposed Method B that started from hydroxymethylcyclohexane 3. Each student believed that their proposed method is better than the other. (Scheme below) Ph THF A Ph Ph B H₂SO4 100 °C 3 OH (iii) In analysing both these methods, are there other possible alkene products other than methylenecyclohexane 2? Use mechanistic details to support your answer.arrow_forward3. It is required to introduce a halogen group to a five membered ring, thiophene. Discuss the reaction mechanism involved in the reaction by selecting a suitable halogen group and analyze why a particular substituted product obtained after the reaction is predominant over the other possible product(s) with the help of reactions. To meet the ever increasing demand of global population, the demand for textile products and consumption of dyes by these industries is increasing.arrow_forward
- In cells, vitamin C exists largely as its conjugate base X. X is anantioxidant because radicals formed in oxidation processes abstract thelabeled H atom, forming a new radical that halts oxidation. Draw thestructure of the radical formed by H abstraction, and explain why this Hatom is most easily removed.arrow_forwardChemistry 3. Complete the following reaction scheme. Give all product(s) and indicate major or minor and any relevant stereochemistry. (а) NaOEt heat (b) heat (c) Br NaCN DMF (d) (e) OH H,SO,/H,PO, heat (f) HBr (g) 1) BH. THE 2) H,0, OH (h) 1) Hg(OAC), H-0 2) NaBH, (i) KMNO, NaOH Coldarrow_forward(a) Complete the following sequence of reactions (i.e., give the structure for compounds A and B), giving structural details of all key intermediates. heat & A COOCH3 H₁₂ Ni (b) Provide the bond line structures for the pair of compounds used for the Diels-Alder synthesis of the compound shown below. O B COOCH3 (c) Draw the two major products obtained when (3E,5Z)-2,2,3,6,7-pentamethylocta-3,5-diene reacts with HBr at low and high temperatures. Label the products as the kinetic or thermodynamic product AND, if applicable, use dashes and wedges to show the correct stereochemistry in the obtained products.arrow_forward
- O PRACTICE PROBLEM 8.14 Starting with any needed alkene (or cycloalkene) and assuming you have deuterioace- tic acid (CH3CO,D) available, outline syntheses of the following deuterium-labeled compounds.s el en olad lo nohibbs ad CH3 (a) (CH3)2CHCH2CH,D (b) (CH3),CHCHDCH3 (c) (+ enantiomer) (d) Assuming you also have available BD3:THF and CH3CO2T, can you suggest a synthesis of the following? hab erl (+ enantiomer)he imo (nwond-ben) CH3 H. (asoholea)arrow_forwardNH₂ NHỊCH, tipy (a) acrylamide Diamines are used as the building blocks for the synthesis of pharmaceuticals and agrochemicals. In the above synthesis, draw the structure of the product (b). ChemDoodle 1. LIAIH 2. H₂O • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Include cationic counter-ions, e.g., Na* in your answer, but draw them in their own sketcher. OF (b) 6arrow_forwardWrite the bond-line presentations for each compound shown below; consider them as acids and write the conjugate base of each of them- 2. Bond-line structure of a given compound Conjugate base bond-line structure (CH3)2CCHCOOH cis-stereoisomer HCCCHCHCH3 [(Et);NH]* [(i-Pr)2CHOH2]*arrow_forward
- Rank the compounds in each group in order of increasing reactivity in electrophilic aromatic substitution: (a) C6H6, C6H5Cl, C6H5CHO, C6H5OCH3; (b) C6H5CH3, C6H5NH2, C6H5CH2NH2, C6H5CONH2.arrow_forwardIllustrate the resonance effect of the methoxy group -OCH3, on the structure of the benzene ring. Draw all the oissuvke resonance forms of methoxybenzene, including the hybrid Based on the structures, explain how the presence of the -OCH3 group affects: (i) the reactivity of the benzene ring towards electrophilic attack (ii) the orientation or point of attack of an incoming electrophilic reagent on the benzene ring.arrow_forward(a) When (Z)-3-methylhex-3-ene undergoes hydroboration–oxidation, two isomericproducts are formed. Give their structures, and label each asymmetric carbon atomas (R) or (S). What is the relationship between these isomers?(b) Repeat part (a) for (E)-3-methylhex-3-ene. What is the relationship between the productsformed from (Z)-3-methylhex-3-ene and those formed from (E)-3-methylhex-3-ene?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





